INTRODUCTION
Rapid advancements in the fields of electrophysiology (EP) and interventional cardiology have increased the demand for anesthesia services.1,2 These procedures have grown in complexity and often involve the care of acutely sick patients with multiple comorbidities including advanced cardiac and pulmonary disease. Providing anesthesia for patients undergoing these procedures at an off-site location can be challenging where the environment and equipment may be unfamiliar, space is limited, and with physical barriers between the anesthesia professional and the patient. Analysis of the ASA closed claims database indicates a significant number of injuries occur in the cardiology suite (EP and catheterization lab), second only to the gastroenterology lab.3 Understanding and preparing for the inherent challenges of providing anesthesia in these areas may enhance patient safety.
PREOPERATIVE EVALUATION
Preprocedural evaluation should include a thorough history and physical; review of allergies, specifically an allergy to iodinated contrast; and medication reconciliation. Special attention should be paid to anticoagulants and heart failure regimens. With the exception of ablation procedures, beta-blockers and antiplatelet medications are typically continued in the periprocedural setting.2,4 If a preoperative anesthesia clinic is available, additional evaluation of certain high-risk patients or procedures may be warranted (Table 1).
Table 1: High-Risk Patient Factors That May Warrant Preprocedural Anesthesia Evaluation Prior to Their Off-Site Cardiology Procedure
As these patients are commonly followed by a cardiologist, there may be an extensive workup already completed. A 12-lead electrocardiogram, echocardiogram, and cardiac monitoring report may be available for review. If the patient has a cardiovascular implantable electronic device, the anesthesia professional should review the manufacturer, current settings, indication for placement, and whether the patient is pacer-dependent. The most recent Practice Advisory from the American Society of Anesthesiologists in 2020 did not reach a consensus on the time frame at which a device interrogation should be completed prior to an elective procedure, though the report stated that a majority of ASA members and consultants recommend interrogation 3–6 months prior to the planned procedure.5
Preoperative laboratory testing varies depending on the type of intervention and risk for bleeding. Labs may include a complete blood count, type and screen, coagulation studies, and basic metabolic panel, particularly if contrast is to be used.
ELECTROPHYSIOLOGY LAB
Regardless of the ablation technique employed, complications may arise and need to be addressed immediately as decompensation can be rapid. The most common complication is related to vascular access injuries followed by cardiac perforation/tamponade.6 Perforation needs to be emergently treated by immediate reversal of anticoagulation and pericardiocentesis. The anesthesia professional should be prepared to rapidly administer blood products and start vasopressor infusions when necessary. If hemodynamic collapse ensues and transport to the operating room for a surgical intervention is required, preprocedural planning between the anesthesia and electrophysiology teams on the logistics of transporting an unstable patient will save valuable time. Other potential periprocedural complications include cerebrovascular accident, heart block, pulmonary edema, phrenic nerve palsy, esophageal perforation, and, rarely, pulmonary hemorrhage.6-8
Large-bore intravenous lines, arterial catheters, and/or central catheters should be placed prior to the start of the procedure, as once the patient is draped, it can be impossible to access the patient if an emergency arises. Arterial access is preferred in patients where hemodynamic instability is anticipated or the procedure will be long in duration. Often times, arterial pressure monitoring is obtained by the electrophysiologist as part of the procedure; however, it should be noted the waveform may dampen and become inaccurate if the lumen of the arterial sheath is occluded by a device. Many anesthesia professionals may consider obtaining their own invasive blood pressure monitoring in order to avoid this pitfall and as a way to follow arterial blood gases throughout the procedure. A preprocedural discussion with the electrophysiologist concerning these issues is essential in avoiding blood pressure monitoring difficulty.
Further complicating the anesthesia care are large pieces of equipment for cardiac mapping and fluoroscopic imaging that serve as a physical barrier between the anesthesia team and the patient’s airway. Additionally, the operating table and fluoroscopic C-arm are controlled by the electrophysiologist, which may result in accidental dislodgment of the breathing circuit, intravenous lines, and monitors. These circumstances should be anticipated and extensions added to lines.
CATHETER-BASED ABLATIONS
Catheter ablations are a mainstay treatment option for supraventricular tachycardia (SVT), atrial flutter (AFL), atrial fibrillation (AF), and ventricular tachycardia (VT) with the goal of creating a transmural lesion that permanently eradicates arrhythmogenic cardiac tissue without causing collateral injury to adjacent structures. Shorter duration procedures such as SVT and AFL ablations can be performed without an anesthesia professional using moderate sedation by a qualified nurse under the supervision of the electrophysiologist. However, more complex procedures that require significant time for mapping and ablating may best be performed with an anesthesia professional under monitored anesthesia care or general anesthesia.
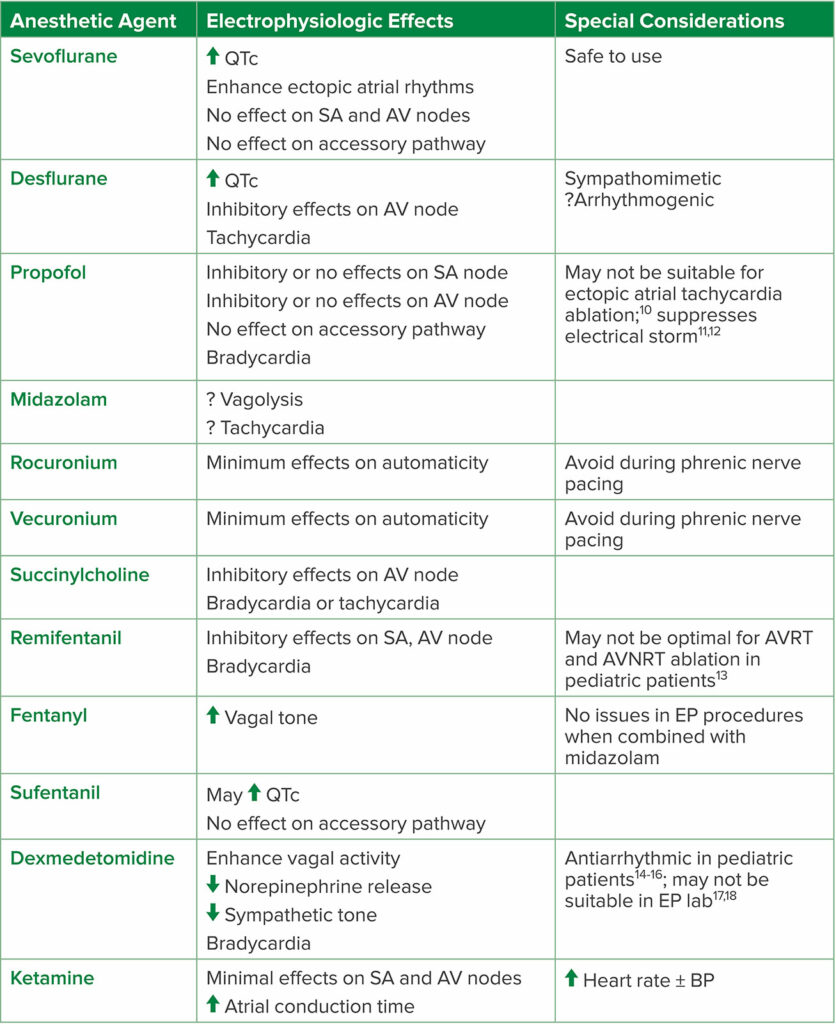
It should also be noted many commonly administered anesthetic drugs may suppress arrhythmia inducibility; therefore, preprocedural discussion with the electrophysiology team is important in determining the appropriate anesthetic for the patient (Table 2).9
Table 2: Anesthetic Agents and Their Electrophysiologic Effects
Abbreviations: AV, atrioventricular; AVNRT, atrioventricular nodal reentrant tachycardia; AVRT, atrioventricular reentrant tachycardia; BP, blood pressure; EP, electrophysiology; SA, sinoatrial
Adapted from the Journal of Cardiothoracic and Vascular Anesthesia, Vol 32, Issue 4. Satoru Fujii, Jian Ray Zhou, Achal Dhir, Anesthesia for Cardiac Ablation, Pages 1892–1910, Copyright (2018), with permission from Elsevier.
Radiofrequency (RF) ablation
Radiofrequency (RF) ablation of the endocardium is the most widely used technique for a variety of arrhythmias where electromagnetic energy is converted to thermal energy resulting in an irreversible thermal injury to myocardial tissue. Typically, active cooling through saline administration at the tip of the catheter is performed in order to prevent collateral injury from excess temperatures at the electrode-tissue interface.19 A useful intervention when an RF procedure involves the left atrium (i.e., AF ablation) is to place an esophageal temperature probe which allows continuous temperature monitoring minimizing this risk to adjacent structures such as the esophagus. Maintaining an esophageal temperature ≤38.5°C may be associated with a decrease in esophageal injuries such as ulceration and left atrial-esophageal fistula formation.20 Also, active cooling may result in several liters of saline being administered by the electrophysiologist over the course of the procedure and must be taken into account when assessing overall fluid balance. This is especially true for patients with poor ventricular function.
Cryoballoon ablation
Cryoballoon ablation is a newer technology, mainly used in the treatment of AF, that freezes the endocardium resulting in impaired propagation of aberrant electrical signals. A balloon-tipped catheter is inserted into a pulmonary vein that, when inflated, circumferentially freezes the surrounding tissue. One technical consideration for the anesthesia professional is the avoidance of muscle relaxants because phrenic nerve stimulation is often employed. Phrenic nerve palsy is one of the most common complications after cryoballoon ablation.21
Epicardial Ablation
An epicardial approach to ablation may be employed for certain ventricular arrhythmias and as part of a hybrid surgical-catheter technique for AF. The hybrid approach is a relatively new technique where both the epicardium and endocardium are treated, which may provide some added benefit in the treatment of AF by combining both surgical (epicardial) and catheter (endocardial) approaches.22 These epicardial procedures are exclusively performed under general anesthesia. If hypotension is encountered during an epicardial approach, unique complications that should be suspected include injury to a coronary artery and intra-abdominal bleeding.7
CATHETERIZATION LABORATORY
Transcatheter Aortic Valve Replacement
Over the last several years, the indications and anesthetic considerations for Transcatheter Aortic Valve Replacement (TAVR) have evolved. Once only indicated for patients with severe, symptomatic aortic stenosis (AS) for whom surgical aortic valve replacement was deemed too high risk, approval was recently expanded for use in low-risk, symptomatic patients with AS.23,24 Furthermore, TAVR is being evaluated for asymptomatic patients with severe AS.
There are currently two TAVR systems used in the United States; the Edwards Sapien valves and the Medtronic CoreValve family of devices. The Sapien valve is a low profile, balloon expandable valve that cannot be repositioned following deployment, whereas the CoreValve family of valves is self-expanding, higher profile, and can be partially recaptured and repositioned for optimal placement.
Use of the Sapien valve or performance of balloon aortic valvuloplasty prior to valve deployment requires rapid ventricular pacing (160–220 beats/minute) via a temporary transvenous pacer. This minimizes blood flow in the left ventricular outflow tract thereby reducing the risk of valve migration during deployment.25 Rapid pacing and subsequent hypotension may not be well tolerated by patients with aortic stenosis, but this situation is usually transient; use of vasopressors such as phenylephrine or norepinephrine should be considered to treat hypotension only if it is persistent as rebound hypertension may develop after pacing ceases.
While overall mortality for TAVR remains low at 1–4%, complications can lead to significant morbidity. The majority of complications are identified intraoperatively and include vascular injury (4.2%), aortic dissection (0.2%), ventricular perforation leading to tamponade (1%), valve malposition and malfunction (0.3%), annular rupture (0.4%), stroke, myocardial infarction, and high degree atrioventricular nodal block requiring permanent pacemaker (8.8%).26
The most common approach for device placement is transfemoral (95%). Other approaches include subclavian/axillary, transaortic, transapical, transcaval, and transcarotid. The transfemoral approach has the benefit of minimal discomfort for the patient and minimal sedation requirements. As technology becomes more sophisticated, and interventionalists become more skilled, utilizing mild-moderate sedation for TAVR has grown in popularity. Recent data shows that benefits include less vasopressor use, a modest decrease in in-hospital mortality, shorter hospital length of stay, and more frequent discharge to home.27 When utilizing sedation with local anesthesia, device placement is confirmed with fluoroscopy and transthoracic echocardiography (TTE).
If transesophageal echocardiography (TEE) is preferred over TTE or when a percutaneous transfemoral approach is not feasible, often due to inadequate iliofemoral vasculature, or a surgical cutdown for vascular repair is required, general anesthesia with an endotracheal tube is utilized. Benefits of general anesthesia include a quiet surgical field, complete control of the airway and early recognition of surgical complications with TEE.
Regardless of anesthesia type, invasive blood pressure monitoring is recommended. This can be accomplished via a radial arterial line or by transducing the arterial sheath used by the interventionalist for aortography. Large bore, peripheral IV access and immediate access to cross-matched blood is also recommended.
Transcatheter Mitral Valve Repair or Replacement
Transcatheter mitral valve repair (TMVr) may be considered for patients with symptomatic, moderate-severe, or severe mitral regurgitation for whom surgical valve repair is considered too high risk. The MitraClip device (Abbott Vascular-Structural Heart, Menlo Park, CA) is currently the only device with FDA approval and is performed in a cardiac catherization lab or hybrid operating room. The MitraClip device is a leaflet repair device and is modeled after the surgical Alfieri stitch which creates an edge-to-edge repair and double orifice mitral valve, thereby reducing the degree of mitral regurgitation.28
When performing transcatheter leaflet repair, transfemoral venous cannulation is obtained by the proceduralist. Using real-time fluoroscopic and TEE guidance, the device is directed across the intraatrial septum, through the left atrium and across the mitral valve into the left ventricle. Both two-dimensional and three-dimensional TEE imaging are imperative to accurately position the device. Immediately following MitraClip release, the degree of mitral regurgitation and iatrogenic stenosis are assessed with TEE. If placement is suboptimal, the clip can be retrieved, repositioned, or removed. More than one clip can also be used to reduce the amount of regurgitation, if necessary.29
General anesthesia with an endotracheal tube is recommended, given the importance of TEE for device placement. Radial arterial access is typically obtained by the anesthesia professional for close hemodynamic monitoring and blood draws. Frequent lab draws may be required to achieve the desired level of anticoagulation. If radial artery access is challenging, other arterial access sites may be utilized. Central venous line placement is not typically necessary, though large bore IV access is recommended due to the risk of emergent conversion to open surgical repair. Cross-matched blood should be available in the procedure room.29
Complications of TMVr include partial clip detachment or embolization, tamponade, bleeding at access sites, and iatrogenic mitral stenosis. It is important to note that TMVr may result in an iatrogenic atrial septal defect at the site of septal puncture. If a shunt is noted, all intravenous lines should be closely evaluated for air to prevent stroke.
Alternatively, and less commonly performed, transcatheter mitral valve replacement (TMVR) is FDA approved for high-risk patients that have a failing mitral valve previously replaced or repaired with a bioprosthetic valve or annuloplasty ring, respectively. The Edwards Sapien 3 or Sapien 3 Ultra, which are designed for TAVR, are used in these patients for valve-in-valve or valve-in-ring replacement. Some institutions are also using TAVR valves in an off-label manner to treat end-stage, refractory native mitral valve disease. TMVR technology is still evolving and its use has been limited due to poor outcomes. Similar to TMVr, general anesthesia is typically used for TMVR due to the necessary use of TEE.
ADDITIONAL OFFSITE CARDIAC PROCEDURES
Diagnostic Transesophageal Echocardiography
TEE is utilized to better visualize cardiac structures that are not well visualized by TTE. While routine use of TEE is not appropriate, as TTE carries little to no risks and is often times diagnostically adequate, there are several clinical situations where TEE is preferred. Clinical indications for TEE may include valvular pathology and surgical planning, urgent assessment of acute aortic pathology (i.e., aortic dissection), diagnosis of infectious endocarditis, and prior to nonemergent direct-current cardioversion (DCCV) or ablation to assess for intracardiac thrombus.
Diagnostic TEE is typically performed with moderate sedation. Apnea should be avoided. Careful topicalization of the pharynx with lidocaine can be used to decrease the amount of sedation required. Topicalization with benzocaine has fallen out of favor due to the risk of methemoglobinemia. Intravenous glycopyrrolate can also be used to minimize oral secretions.30 Stimulation associated with TEE probe insertion can be mitigated with a propofol bolus of 0.25–0.5 mg/kg. Following insertion, the degree of stimulation quickly decreases, and moderate sedation can be achieved with a low dose propofol infusion or incremental propofol boluses. Propofol has the benefit of rapid onset and metabolism, and minimal residual effects following the procedure.28 Alternatively, a dexmedetomidine bolus of 0.5–1 mcg/kg over ten minutes and/or an infusion of 0.2–1 mcg/kg/hour can be used in conjunction with adequate airway topicalization.
In certain high-risk patient populations, such as those with a difficult airway, high aspiration risk, impaired neurologic status or those with airborne precautions, such as COVID-19, general anesthesia with an endotracheal tube may be warranted. As TEE is an aerosolizing procedure, its elective use should be avoided in patients with COVID-19 unless the findings will change clinical management.
Although TEE is a generally safe procedure, complications such as laryngospasm, aspiration, pharyngeal injury, perforated viscus, and hemorrhage do occur. Initial treatment for such adverse events is typically endotracheal intubation and resuscitation.
Direct-Current Cardioversion (DCCV)
DCCV is usually a short procedure requiring a rapid onset and offset of anesthesia. Following application of standard ASA monitors and capnography, a 0.25–0.5 mg/kg propofol bolus is administered such that the patient is not responsive to tactile or verbal stimulation. Apnea should be avoided. Once deep sedation is confirmed, the electrical shock can be delivered. Patients undergoing DCCV may have low cardiac output, slow circulation time, and delayed onset of induction medications which can lead to oversedation. Medications to treat hypotension and/or bradycardia, such as phenylephrine; ephedrine; and glycopyrrolate or atropine, should be readily available.28,31 Pre-procedure external defibrillation pads should be placed in the event of post-DCCV asystole and extrinsic pacing is required. If a patient has an implantable electronic cardiac device, such as a pacemaker or defibrillator, the device should be interrogated immediately following external cardioversion or defibrillation.8
CONCLUSIONS
As cardiac interventions become more sophisticated and less invasive, anesthesia professionals are tasked with providing safe medical care in a wide variety of locations, often far removed from the operating room. Additionally, the patients undergoing such procedures have complex medical histories and are more acutely ill. As an integral component of the care team, it is imperative that the anesthesia professional is familiar with the challenges of off-site procedures, understands the procedure itself, and can anticipate pitfalls so as to provide safe patient care.
REFERENCES
- Gaitan BD, Trentman TL, Fassett SL, et al. Sedation and analgesia in the cardiac electrophysiology laboratory: a national survey of electrophysiologists investigating the who, how, and why? J Cardiothorac Vasc Anesth. 2011;25:647–659.
- Anderson R, Harukuni I, Sera V. Anesthetic considerations for electrophysiologic procedures. Anesthesiol Clin. 2013;31:479–489.
- Metzner J. Posner KL, Domino KB. The risk and safety of anesthesia at remote locations: the US closed claims analysis. Curr Opin Anaesthesiol. 2009;22:502–508.
- Shook D, Evangelista K. Anesthetic considerations for electrophysiology, interventional cardiology, and transesophageal echocardiography procedures. In: Post TW, ed. UpToDate. UpToDate; 2021. Accessed September 9, 2021.
- Practice Advisory for the Perioperative Management of Patients with Cardiac Implantable Electronic Devices: Pacemakers and Implantable Cardioverter–Defibrillators 2020: An Updated Report by the American Society of Anesthesiologists Task Force on Perioperative Management of Patients with Cardiac Implantable Electronic Devices. Anesthesiology. 2020;132:225–252.
- Gupta A, Perera T, Ganesan A, et al. Complications of catheter ablation of atrial fibrillation: a systematic review. Circ Arrhythm Electrophysiol. 2013;6:1082–1088.
- Kumareswaran R, Marchlinski FE. Practical guide to ablation for epicardial ventricular tachycardia: when to get access, how to deal with anticoagulation and how to prevent complications. Arrhythm Electrophysiol. Rev. 2018;7:159–164.
- Haines DE, Beheiry S, Akar JG, et al. Heart Rhythm Society expert consensus statement on electrophysiology laboratory standards: process, protocols, equipment, personnel, and safety. Heart Rhythm. 2014 Aug;11:e9–51.
- Fujii S, Zhou JR, Dhir A. Anesthesia for cardiac ablation. J Cardiothorac Vasc Anesth. 2018;32:1892–1910.
- Lai LP, Lin JL, Wu MH, et al. Usefulness of intravenous propofol anesthesia for radiofrequency catheter ablation in patients with tachyarrhythmias: infeasibility for pediatric patients with ectopic atrial tachycardia. Pacing Clin Electrophysiol. 1999;22:1358–1364.
- Burjorjee JE, Milne B. Propofol for electrical storm: a case report of cardioversion and suppression of ventricular tachycardia by propofol. Can J Anaesth. 2002;49:973–977.
- Mulpuru SK, Patel DV, Wilbur SL, et al. Electrical storm and termination with propofol therapy: a case report. Int J Cardiol. 2008;128:e6–8.
- Niksch A, Liberman L, Clapcich A, et al. Effects of remifentanil anesthesia on cardiac electrophysiologic properties in children undergoing catheter ablation of supraventricular tachycardia. Pediatr Cardiol. 2010;31:1079–1082.
- Hammer GB, Drover DR, Cao H, et al. The effects of dexmedetomidine on cardiac electrophysiology in children. Anesth Analg. 2008;106:79–83.
- Chrysostomou C, Sanchez-de-Toledo J, Wearden P, et al. Perioperative use of dexmedetomidine is associated with decreased incidence of ventricular and supraventricular tachyarrhythmias after congenital cardiac surgery. Ann Thorac Surg. 2011;92:964–972.
- Herr DL, Sum-Ping STJ, England M. ICU sedation after coronary artery bypass graft surgery: Dexmedetomidine-based versus propofol-based sedation regimens. J Cardiothorac Vasc Anesth. 2003;17:576–584.
- Mittnacht AJC, Dukkipati S, Mahajan A. Ventricular tachycardia ablation: a comprehensive review for anesthesiologists. Anesth Analg. 2015;120:737–748.
- Hayman M, Forrest P, Kam P. Anesthesia for interventional cardiology. J Cardiothorac Vasc Anesth. 2012;26:134–147.
- Houmsse M and Daoud EG (2012) Biophysics and clinical utility of irrigated-tip radiofrequency catheter ablation. Expert Review of Medical Devices. 9:1,59–70.
- Singh SM, d’Avila A, Doshi SK, et al. Esophageal injury and temperature monitoring during atrial fibrillation ablation. Circ Arrhythm Electrophysiol. 2008;1:162–168.
- Tokuda, M., Yamashita, S., Sato, H. et al. Long-term course of phrenic nerve injury after cryoballoon ablation of atrial fibrillation. Sci Rep 11, 6226 (2021).
- Driver K, Mangrum JM. Hybrid approaches in atrial fibrillation ablation: why, where and who? J Thorac Dis. 2015;7:159–164.
- Mack MJ, Leon MB, Thourani VH, et al. Transcatheter aortic-valve replacement with a balloon-expandable valve in low-risk patients. N Engl J Med. 2019;380:1695–1705.
- Coylewright M, Forrest J, McCabe J, Nazif T. TAVR in low-risk patients: FDA approval, the new NCD and shared decision-making. JACC. 2020;75:1208–1211.
- Novak T, Parulkar S. The anesthesia professional’s role in patient safety during TAVR (transcatheter aortic valve replacement). APSF Newsletter. 2017;31:73–75.
- Dalby M, Panoulas V. Transcatheter aortic valve replacement: complications. In: Post TW, ed. UpToDate, UpToDate, 2021. Accessed October 1, 2021.
- Butala NM, Chung M, Secemsky EA, et al. Conscious sedation versus general anesthesia for transcatheter aortic valve replacement: variation in practice and outcomes. J Am Coll Cardiol Intv. 2020;13:1277–1287.
- Faillace R, Kaddaha R, Bikkina M, et al. The role of the out-of-operating room anesthesiologist in the care of the cardiac patient. Anesthesiology Clin. 2009;27:29–46.
- Gregory SH, Sodhi N, Zoller JK, et al. Anesthetic considerations for the transcatheter management of mitral valve disease. J Cardiothorac Vasc Anesth. 2019;33:796–807.
- Hahn R, Abraham T, Adams M, et al. Guidelines for performing a comprehensive transesophageal echocardiographic examination: recommendations from the American Society of Echocardiography and the Society of Cardiovascular Anesthesiologists. J Am Soc Echocardiography. 2013;26:921–964.
- Lu F, Lin J, Benditt D. Conscious sedation and anesthesia in the cardiac electrophysiology laboratory. J Cardiovasc Electrophysiol. 2013;24:237–245.



Leave a Reply
You must be logged in to post a comment.