My patient had very little urine and what did appear was very concentrated. Therefore, I pushed forward with a presumptive diagnosis of hypovolemia as at least one major cause of his ongoing hypotension. I therefore began an aggressive fluid resuscitation with lactated ringers. After four liters given over about 1.5 hours, there was little if any change in any of the patients parameters. In fact, there was almost no urine output. At this point I gave 250 mL of 5% albumin. Blood loss was not substantial, however, there was an approximate 3 liter loss of fluid from the peritoneal cavity of presumptive ascites. It was noted that the patient had a visibly cirrhotic liver. Large volume paracentesis is known to lead to arteriolar vasodilation and an increase in cardiac output. It is widely recognized that paracentesis induced circulatory dysfunction can lead to significant morbidity in patients with liver disease. Management recommendations include post removal treatment with 125 mL of 5% albumin volume replacement per liter of ascitic fluid removed (if more than 5L is removed), and consideration of vasopressors such as terlipressin for continued hypotension.
Recently, a large number of studies have attempted to determine the best way to manage volume status in patients with or at risk for hypotension. Maintaining perfusion pressure and thus DO2 to tissues is critical to avoid organ damage secondary to hypoxic injury. However, some studies have also shown harm to patients associated with overhydration. In Anesthesiology, a study [1] in radical cystectomy patients found that the rate of complications was 52% in a group of patients who received a low volume of LR vs a high volume of LR where the rate of complications was 73%. The low volume group received LR at 1 mL/kg/hr until completion of cystectomy and then 3 mL/kg/hr until the end of surgery plus low dose norepinephrine. The high volume group received 6 mL/kg/hr plus 250 mL fluid boluses as need during surgery. Post op fluid therapy was similar between groups. The rationale for using 6 mL/kg/hr in this study likely came from traditional teaching in textbooks such as Stoelting where we are taught that 6 to 8 mL/kg/hr should be used to maintain hydration in patients undergoing major abdominal surgery [2]. In 2003, Brandstrup et al was able to show a reduction in complications by using a low volume algorithm in elective colorectal surgery. In that study, the high volume group received 7 mL/kg/hr x 1 hour, then 5 mL/kg/hr x 2 hours, and then 3 mL/kg/hr thereafter. Blood loss was replaced by NS up to an EBL of 500 mL, and then colloid up to an EBL of 1500 mL. The restrictive group only received 500 mL D5W and 6% Hetastarch for blood loss up to 1500 mL. The standard group received 5.4 L overall and the restrictive fluid group received only 2.7L. Cardiopulmonary complications were reduced in the restrictive fluid group from 24% to 7% and the incidence of tissue healing complications was reduced from 31% to 16%. In 2018 a large international RCT trial compared a restrictive fluid therapy regimen to a liberal strategy in patients having major abdominal surgery. In the liberal group crystalloid was given at 10 ml/kg during induction of anesthesia, followed by an 8 ml/kg/hr infusion until the end of surgery. Post op patients received 1.5 ml/kg/hr for 24 hours. The restrictive regimen was designed to provide a net zero fluid balance. An infusion of crystalloid at a dose of 5 ml/kg/hr was administered until the end of surgery. Post fluids were given at 0.8 ml/kg/hr. During surgery, colloid or blood was given to replace blood loss in a 1:1 ratio. The primary outcome measure was rate of disability free survival at 1 year. There were no differences between groups for this outcome. The secondary outcome was AKI which occurred in 8.6% of the restrictive group and 5% of the liberal group <0 .001=”” 1.7l.=”” 13.6=”” 16.5=”” 1=”” 30-day=”” 3=”” 3l=”” 40=”” a=”” abdominal=”” about=”” administered=”” administration=”” aki.=”” an=”” and=”” anesthesiologists=”” another=”” appropriate=”” as=”” associated=”” association=”” average=”” be=”” between=”” both=”” but=”” by=”” can=”” case.=”” challenges=”” characterized=”” complications=”” concluded=”” consistently=”” costs=”” crystalloid=”” defined=”” difference=”” differences=”” during=”” enough=”” equates=”” estimates.=”” find=”” fluid=”” for=”” found=”” group=”” hand=”” have=”” having=”” he=”” highest=”” his=”” hour=”” in=”” incidence=”” intraoperatively=”” l=”” length=”” less=”” liberal=”” lowest=”” major=”” moderate=”” mortality=”” n=”” nbsp=”” ndeed=”” needle=”” observational=”” observed=”” of=”” on=”” optimal=”” other=”” outcome.=”” outcomes=”” p=”” patients=”” postoperative=”” quintile=”” received=”” regimen=”” relief=”” respiratory=”” restrictive=”” revealed=”” same=”” second=”” secondary=”” sick=”” significantly=”” small=”” stay.=”” strategy=”” study=”” surgery.=”” surgery=”” textbook=”” than=”” that=”” the=”” therapy=”” there=”” this=”” threading=”” to=”” traditional=”” trial=”” u-shaped=”” underscores=”” volume=”” volumes=”” was=”” we=”” were=”” whereas=”” while=”” with=””>Traditional thresholds for intraoperative oliguria do not predict acute kidney injury (AKI). In a meta analysis [4] done in 2016 of 28 trials including both surgical and critically ill patients, less renal dysfunction was noted in patients receiving goal directed fluid therapy without the use of oliguria to guide fluid administration. Another meta analysis by Cochrane concluded, “The balance of current evidence does not support widespread implementation of this approach to reduce mortality but does suggest that complications and duration of hospital stay are reduced.” Specifically they showed that GDT reduced the rate of renal failure, respiratory failure and wound infection. Typically, goal directed therapy for fluid management has relied upon esophageal doppler technology to estimate stroke volume. This technology is not readily available however. Technology to analyze arterial waveforms to estimate pulse pressure variation (PPV) and stroke volume variation (SVV) as as well as systolic pressure variation (SPV) has been developed and is making inroads into many operating rooms. However, these technologies are not wide spread and easily applied. In 2012 Thiele et al published a study showing that anesthesia providers were able to make correct diagnostic decisions in 96% of situations using a simple visual “eyeball” review of systolic blood pressure waveforms attempting to estimate systolic blood pressure variation. This provides evidence that in patients who have an arterial line in place, anesthesiologists who do not have access to higher level arterial waveform analysis technology can be “good enough” to determine which patients are fluid responsive.
Goal directed therapy using stroke volume variation (SVV) or pulse pressure variation (PPV) and arterial waveform analysis has been studied intensely in the last decade. The general concept relates to an attempt to determine if a patient who is hypotensive would respond to a fluid challenge (best is 3 to 4 mL/kg) by increasing stroke volume by about 10 to 15%. During mechanical ventilation there is a rise in pleural pressure during the inspiratory phase which impedes blood return to the right atrium and thus the right ventricle. At the same time as right heart preload is decreased, right heart afterload is increasing during the inspiratory phase in conjunction with increased pleural pressure. However, left ventricular preload increases while left ventricular afterload decreases. This transient alteration in right heart and left heart preload and afterload leads to a decrease in LV output a few heartbeats after completion of mechanical insufflation (or expiratory phase of mechanical ventilation). The changes in the RV and LV stroke volume with each mechanical breath are larger on the steep compared with the flat portion of the Frank Starling curve for four main reasons: 1) the SVC is more collapsible in hypovolemia, 2) the inspiratory increase in right atrial pressure is greater in hypovolemic states secondary to the greater transmission of pleural pressures to the more compliant right atrium, 3) the effect of mechanical inspiration on RV after load is greater because of higher trans alveolar pressures in the setting of hypovolemia, and 4) the ventricles are more sensitive to preload when they are operating on the steep portion of the frank starling curves. To validate this Michard et al studied 40 patients with sepsis on mechanical ventilation. This study found higher variations in pulse pressure (24% vs 7%) in a group of patients who responded to volume expansion (defined by a 15% increase in CI) vs those that did not. They showed that if pulse pressure varied by more than 13% there was a 94% sensitivity (low false negative) and a 96% specificity (low false positive) in predicting volume responsiveness [5]. Nine years after this initial study, a meta analysis was completed by Marik et al [5]. This group looked at 29 clinical studies. They found that the area under the ROC curve was 0.94 for PPV and 0.86 for SVV. All other strategies utilized for determining volume status (CVP, global end-diastolic volume index, LVEDA index) performed poorly. They also found a very consistent threshold for defining fluid responsiveness of 12 to 13%. They also noted that there is a gray zone of 9 to 13% where fluid responsiveness cannot be predicted reliably and this ‘gray zone’ may affect up to 25% of patients during general anesthesia. Furthermore, in order to perform arterial waveform analysis using current technology, patients must be in sinus rhythm, be mechanically ventilated with tidal volumes greater than 7 mL/kg and not be receiving vasopressors.
When confronting a patient whose volume status is in question and hypotension is at hand, a detailed conceptual construct is helpful in understanding how to proceed. This construct aids in understanding the mechanisms of how fluid therapy can be harmful or helpful and when. One concept that was previously highlighted in the journal Anesthesiology divides total blood volume into the stressed and unstressed volumes within the body. The ‘unstressed’ volume is that volume of blood that fills the blood vessels without causing a rise in pressure. The ‘stressed’ volume is any additional volume that results in a rise in pressure AND elastic distention of the vessel wall. Therefore, when a clinican administers a fluid challenge, they are aiming to expand the ‘stressed’ volume. Whether the fluid challenge leads to a rise in pressure or not is dependent upon whether it fills the ‘unstressed’ volume or the ‘stressed’ volume. This is dependent upon the overall venous compliance. In 1894, Bayliss and Starling first described the concept of mean systemic filling pressure (Pmsf) in a dog model. This is defined as the pressure in the vascular system when the heart is stopped and there is no blood flow. Pmsf is a critical element in determining venous return along with right atrial pressure and resistance to venous return. The driving pressure for venous return is the pressure gradient between Pmsf and central venous pressure (CVP) which then determines cardiac output.
It should be noted that with the induction of anesthesia, or other physiologic changes the unstressed volume can suddenly increase. A sudden increase in the unstressed volume (see above figure) would then lead to a decrease in the Pmsf and thus reduce venous return and thus cardiac output. In a patient who undergoes a temporary state (i.e. general anesthesia) where the unstressed volume is suddenly increased due to sympathectomy, and this is filled aggressively with fluid, and then thereafter, the unstressed volume returns to its previous state (emergence), the patient could now be in a sudden state of fluid overload as the fluid placed into the unstressed volume is now recruited into the stressed volume per force. Therefore, the above conceptual framework allows clinicians to visualize the potential negative effects of overly aggressive fluid therapy. Conceptually, one might consider what happens to the Pmsf in a patient suffering a vasodilatory state such as sepsis. In this scenario, once again, the unstressed volume is dramatically increased leading to a decrease in Pmsf which will decrease venous return and thus cardiac output. If the treatment modality is only additional fluid to fill the unstressed volume, there is a real risk of too much fluid. Therefore, the correct therapeutic modality to in treating an unnatural increase in the venous capacitance or unstressed volume are vasopressors. Vasopressors in this setting serve to return the unstressed volume back to its “natural” volume, at which time, additional fluid therapy can fill the “stressed” volume leading to an increase in Pmsf which would be clinically observable with an increase in SV or CO leading to improved blood pressure.
The above case represents a perfect example of a patient who had vasoplegia secondary to sepsis and therefore, a very large unstressed volume. In addition, the patient was also likely suffering from a capillary leak syndrome leading to loss of intravascular fluid into the interstitial. This fluid would need to be replaced. However, the vasodilated state required Alpha 1 adrenergic therapy. There is evidence that early fluid therapy will do more than just increase the mean systemic filling pressure (Pmsf) promoting increased right heart filling pressures. Early aggressive fluid therapy in sepsis can also shift the cytokine response towards a more anti-inflammatory balance [8] and is associated with reduced mortality in septic patients [9].
In my patient after about 6 liters of crystalloid I infused 250 mL of human 5% albumin. There is a great deal of controversy related to the type of fluids. Currently, there is no clear evidence that crystalloid (balanced salt solution) leads to greater mortality than colloid. In addition hespan has fallen out of favor because it has been linked to AKI in critically ill adults. Risk of HES-induced renal toxicity depends primarily on the molar substitution. For example, the commonly available Hespan or hydroxylethyl starch 6% has a MW of 600 and degree of substitution of 0.75. This formulation started to fall out of favor initially in 2003 when the FDA required that a new label be applied to 6% hetastarch (HESPAN) that recommended against HESPAN in bypass patients due to concerns related to coagulopathy. Newer HES solutions (tetrastarch 130/0.4) are considered far less toxic to the kidneys. A recent [10] observational study compared a newer HES (130/0.4) to crystalloid and found that HES was not associated with an increased frequency of post op kidney failure. Also, in-hospital mortality and ICU requirements were not different between groups. This was a mixed cohort of elective surgical patients. Another study comparing HES 130/0.4 to 5% albumin found no differences in renal function in a small RCT in patients undergoing cystectomy. Another study of HES 130/0.4 found that this formulation could reduce the inflammatory response in patients undergoing major surgery compared to a purely crystalloid based volume regimen. In larger meta analyses where a large number of different types of starches were compared to crystalloids, the incidence of RRT was greater in the patients receiving starches. In another study using pentastarch (200/0.5) vs LR there was a larger incident of AKI in the starch group. However, on subgroup analysis it was found that patients suffering the negative renal outcome were given a larger than usual volume of pentastarch. Patients given less than 22 mL/kg of pentastarch actually suffered a significantly lower mortality (31% vs 58%) vs high dose penta starch and vs. LR (41% mortality). Furthermore, starches, especially those with a high degree of molar substitution (i.e. 0.75) are associated with a greater risk of bleeding and transfusions as per large meta analyses. Fortunately, once again, lower molar substitution products (tetra starch=0.4) seem to have a smaller effect on hemostasis. Avoiding these products (starches in general) would likely be important in particular in the above case as it was found that the patient had a fairly large amount of ascites upon opening the abdomen with a liver that appeared markedly cirrhotic. Although the patient had normal coagulation parameters prior to surgery with no evidence of decreased liver function, it is possible that the patient had some degree of platelet dysfunction from his liver disease which would be a relative contraindication to any form a starch fluid therapy.
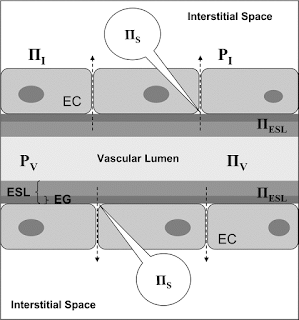
In 2008, a lengthy review article was published on periopeartive fluid management in Anesthesiology [109;723-740]. In addition to being an excellent general review of the literature on perioperative fluid management, the author reviews the importance of the glycocalyx and it’s part in forming the enodthelial surface layer (ESL) (see fig).
This layer is critical in avoiding platelet aggregation, leukocyte adhesion and increased endothelial permeability. The ESL can be damaged by ischemia reperfusion, proteases, TNF-alpha, oxidized LDL lipoproteins, and atrial natriueretic peptide. Therefore, two important things should be noted: 1) overly aggressive fluid hydration even in healthy volunteers may damage the ESL leading to pathologic fluid losses via damage to the ESL. 2) Major surgery and/or patients in a state of sepsis where a large inflammatory response is expected will also have a damaged ESL and pathologic shift of fluids into the interstitial space. Quoting the article from Chappell in Anesthesiology, “Consequently, the primary indication of crystalloids is replacement of fluid losses via (1) insensible perspiration and (2) urinary output. Colloids, by contrast, are indicated to replace plasma deficits due to (2) acute blood loss or (2) protein-rich fluid shifts toward the interstitial space (pathologic type 2 shift).” Therefore, for major surgery, crystalloids would be indicated to replace insensible losses estimated to be about 1 mL/kg/hr (per chappell article). All other fluid needs (i.e. from pathologic type 2 shift (shift of fluid across the endothelial glycocalyx into the interstitium) should be replaced by a colloid solution. Unfortunately, HESPAN is no longer available and HES (130/0.4) is likely to be unavailable as well. 5% albumin is the only choice available in our hospital. Given that the current evidence does not seem to indicate a clear and defined cut off for crystalloid where it is clear that you are killing patients, heavy use of crystalloid is still the mainstay given a lack of alternatives.
Therefore, in the above case, I might have selected a larger 5% albumin dose, maybe 1 liter, but ultimately, given the degree of the fluid needs, I would have been forced to use a fairly large volume of crystalloid. Unfortunately, the vast majority of this crystalloid found its way into the interstitial space (chappell estimates it’s actually 5:1 into the interstitial space not 3:1 as historically taught). Patients with sepsis are also suffering from vasoplegia. Therefore, using the above definition, there unstressed volume will be pathologically increased. An IV vasopressor infusion to decrease the unstressed volume was also critical in facilitating the fluid resuscitation.
In summary, this case represents a patient with a perforation of the large bowel presenting with a significant systemic inflammatory response resulting in shock requiring emergent open abdominal surgery and aggressive fluid therapy. In addition, the patient had co morbidities of liver disease, coronary artery disease, and insulin requiring diabetes. Fluid management was reviewed and an understanding of the mechanisms of how to determine the volume status of patients using objective measures such as PPV to achieve a goal directed infusion of fluids was highlighted. Furthermore, the pros and cons of crystalloid vs colloids was addressed highlighting that crystalloids should in general be reserved to treat ongoing insensible fluid losses while colloids should be utilized for all type II or pathologic fluid losses (including hemorrhage and fluid crossing the endothelial layer into the interstitial space.
1. Wuethrich P, Burkhard F, Thalmann G, Stueber F, Studer U. Anesthesiology. 2014 FEB
2. Stoelting et. al. Basics of Anesthesia, 5th ed. Elsevier – China, p. 349, 2007
3. Brandstrup B et. al. Ann Surg 238: 641, 2003
4. Egal M, Erler NS, de Geus HR, Van Bommel J, Groenevald AB.Anesth Analg. 2016; 122:173-185.
5. Michard F, Boussat S, Chemla D. et al. Am J Resp Grit Care Med. 2000; 162: 134-138.
6. Myles PM et al. NEJM. 2018; 378:2263-2274.
8. Dorresteijn MJ, van Eijk LT, Netea MG, et al. J Endotoxin Res. 2005;11:287–293.
9. Lee SJ, Ramar K, Park JG, et al. Chest. 2014;146:908–915.
10. Pagel JI, Rehm M, Kammerer T, Hulde N, Speck E, Briegel J, Reinholz F, Crispin A, Hofmann-Kiefer KF. Anesth Analg. 2018; 126(6): 1949



Leave a Reply
You must be logged in to post a comment.