Anesthesiology News
The current rate of perioperative cardiac arrest (CA) is estimated to be between 1 and 1,400 to 1,800 anesthetic interventions (0.05%-0.07%).1 The number of CAs directly attributable to anesthesia as a primary causative factor has decreased over the last 5 decades, and is estimated to occur from 0.5 to 1 case per 10,000 interventions (0.005%-0.01%).2-5 Medication overdose, provider inattention, machine malfunction, human error, and respiratory-based events (eg, airway management difficulties) are responsible for most anesthesia-related catastrophies.2-5
Emergency airway management outside the operating room (OR) may be plagued with patient care safety issues. Acute physiologic derangements, cardiopulmonary compromise, and hemodynamic deterioration, coupled with the emergency’s circumstances, insufficient patient preparation, and an abbreviated assessment, provide a high-risk environment for critical airway care. Hemodynamic complications, difficulty with oxygenation and ventilation, esophageal intubation (EI), aspiration, multiple intubation attempts, and the need to deploy multiple airway devices continue to afflict patient safety.6-8
An undesirable yet not uncommon consequence of acute physiologic turmoil and airway management difficulty is complete cardiovascular collapse (CVC) after intubation. CVC would seem to be a comprehensive description for post-intubation CA, but the definition of CVC varies among investigators. A recent publication by Perbet et al6 defined CVC as an arterial systolic blood pressure no more than 65 mm Hg recorded at least once and/or no more than 90 mm Hg lasting at least 30 minutes, following intubation. These hemodynamic alterations occur despite vascular loading with 500 to 1,000 mL of crystalloid and/or colloid solutions and/or the necessity of introducing vasoactive drugs.
Assuming the intraoperative CA rate attributable to anesthesia ranges from 1 in 10,000 anesthetic interventions, with an even smaller proportion directly related to airway management difficulties, the relative risk for CA-NOR appears to be 200- to 400-fold higher or greater than its in-OR counterpart.7-9,12 Over the last several decades, recommendations by several societal bodies involved with airway management (the ASA, Difficult Airway Society, Canadian Airway Focus Group) encouraged the deployment of endotracheal tube (ETT)-verifying devices (end-tidal carbon dioxide [EtCO2]) and advanced airway devices—a bougie catheter, supraglottic airway device, and bronchoscope—which have had an impressive effect on elective and emergent airway management.13 The high rate of airway- and hemodynamic-related complications were the focus that prompted such management recommendations.8 Societal expertise and recommendations were needed to usher in patient safety initiatives to ensure immediate availability of advanced airway management equipment and ETT-verifying devices.
An example of the patient care improvements that were garnered by the implementation of the ASA Guidelines for bedside availability of such airway management adjuncts was studied in an emergency intubation database over 2 time periods: Group 1: 1990 to late 1995 (before/after the ASA Guidelines but prior to their hospital-wide implementation) and Group 2: late 1995 to 2002 (after their implementation).8 Of note, major airway management difficulties contributed to nearly 75% of CA-NOR during the pre-ASA time frame, when the airway team practiced with limited availability of advanced airway adjuncts and EtCO2 verification devices. After the ubiquitous distribution of such airway equipment (ie, Group 2), the CA-NOR rate dropped precipitously, likely due to the reduction in airway management difficulties involved in the unfortunate patients experiencing CA-NOR (Table 1). These safety initiatives afforded a welcome reduction in the overall rate of CA-NOR by 50%.8
| Table 1. Description of Available Airway Management Equipment in Non-OR Areas | |
| Groupa | Equipment Available at Bedside |
|---|---|
| 1 | Conventional equipment only (advanced devices in stock room, available by phone or page) |
| 2 | Conventional (EtCO2 capable) + portable bagb at bedside (advanced devices); difficult airway cartsc in all ICUs, the ED, radiology, the endoscopy suite, the cardiac catheterization lab, high-traffic floor locations |
| 3 | All period 2 equipment + a video laryngoscope (GlideScope Ranger in portable bag, GlideScope GVL via a wheeled pole, Airtraq in bag) |
|
a 1=1990 to late 1995; 2=late 1995 to mid-2006; 3=mid 2006 to 2017.
b Includes a bougie catheter, airway exchange catheters, a variety of supraglottic airway devices (LMA Classic [Teleflex], intubating LMA [Fastrach, Teleflex], LMA Supreme [Teleflex], Combitube [Medtronic]), a transtracheal jet ventilation set, and a Cook Cricothyrotomy Kit.
c Based on recommendations of the ASA’s “Practice Guidelines for Management of the Difficult Airway”13 (includes fiber-optic bronchoscopy).
ED, emergency department; EtCO2, end-tidal carbon dioxide; ICU, intensive care unit; OR, operating room
|
|
Specifically, there was an impressive reduction in CA-NOR related to airway management difficulties. These relatively rudimentary practice additions reduced the incidence of severe oxygen desaturation during airway manipulation by lowering the need for multiple intubation attempts (3+), lowering the incidence of regurgitation and aspiration, reducing the delay in recognizing EI, and limiting suffering from repetitive EI.8
Further enhancements in improving the ability to intubate the trachea using video-assisted laryngoscopy (VAL) have altered the airway management landscape and ushered in improved patient airway care in both elective and urgent circumstances. Although emergency intubation has not been thoroughly researched, the positive impact of VAL with improved successful intubation rates of the difficult airway patient, improved first-pass success, and a reduced incidence of EI are welcome improvements to patient care and airway safety.14-22
The primary goal of the present database review was to learn whether bedside availability of societal-recommended ETT-verifying capabilities and advanced airway equipment is augmented by VAL deployment in reducing the rate of CA-NOR further, especially those CAs attributed to difficult airway management.1-5,8 The secondary goal was to determine whether bradycardia-based CA-NOR would be less common due to the reduction in airway-related complications leading to CA-NOR.
As an ongoing quality improvement project, Hartford Hospital’s emergency intubation database was analyzed to determine the incidence and risk factors contributing to intubation-related CA in the remote non-OR hospital location (NOR) taking place over 3 time periods: 1990 to late 1995 (Group 1), late 1995 to mid-2006 (Group 2), and mid-2006 to 2017 (Group 3) (Table 1). The institution’s investigational review board approved a retrospective review of the ongoing emergency intubation database covering a 329-month period (August 1990 to December 2017). Adult patients (aged 17-101 years) who required intubation outside the OR at Hartford Hospital, a tertiary care, Level 1 trauma center, were included. Tracheal intubations of patients with cardiopulmonary resuscitation (CPR) already in progress were excluded.
The anesthesia airway team was the primary provider of 24/7 airway management coverage for all hospital locations except the ED, and consisted of at least 1 board-certified anesthesiologist working alone or supervising anesthesia trainees. Coverage during the vast majority of the daytime shifts (7 am to 5 pm) was by anesthesia intensivists. The anesthesia team provided second responder coverage for airway assistance in the ED when initiated by a “stat page” for rescue assistance. Airway managers used their clinical judgment to determine the approach to airway management (ie, intubation route, premedications, etc). The respiratory therapy and nursing staff were first responders and provided noninvasive bag-valve-mask oxygen supplementation before arrival of the anesthesia airway team. Ventilation with 100% oxygen by the anesthesia personnel for 2 to 4 minutes before and between prolonged intubation attempts (>30 seconds) was standard practice. From 1990 to late 1995, accessory airway devices and equipment to assist with advanced airway techniques were sparse and not immediately available at the bedside except by acquisition from the anesthesia equipment room, which required telephone call/pager communication and then transport. In late 1995, a 3-tier equipment deployment was organized to provide bedside airway equipment to reflect published recommendations:
- First, an airway tackle box with conventional intubation equipment, including EtCO2detection equipment, was distributed to 62 clinical locations throughout the institution.
- Second, transportable airway bags, stocked with invasive and noninvasive accessory devices, were located at anesthesia work areas and readily available to be carried to the bedside after an “anesthesia stat” was announced.
- Third, difficult airway carts stocked with a wide variety of airway equipment (based on the ASA recommendations), including flexible fiber-optic bronchoscopes, were deployed in high-traffic non-OR areas, for example, the ICU (5 locations), the ED, the radiology department (2 locations), cardiac catheterization lab, endoscopy suite, and selected high-traffic floor locations, etc. After the mid-2006 acquisition of VAL equipment, the GlideScope Ranger (Verathon) was stocked in the airway bags, and the GlideScope GVL (Verathon) was transported via a wheeled pole assembly to the patient’s bedside (Table 1).
After intubation, the airway team completed a data form that outlined patient demographics, procedural details, hemodynamic alterations, airway-related mishaps, and complications. This was replaced in late 2009 by an intubation form recording the same information, but was used for both billing and medical record documentation. Demographics included the patient’s age, sex, medical history, and primary diagnosis necessitating tracheal intubation. Procedural data included patient location, route of intubation, level of training, number of attempts, medications administered for patient preparation, pre- and post-intubation hemodynamic data, airway devices used, and complications. The author reviewed the data form and completed any missing information by reviewing the medical record and interviewing the airway team, and then entered the data into the computer-based database.
Patient Demographics
All cases of CA from 1990 through 2017 were identified from Hartford Hospital’s computerized database to estimate the incidence of intubation-related CA. A total of 21,462 completed emergency intubation forms were entered into the database, of which 2,825 were intubations for preexisting CAs. Thus, a total of 18,637 emergency intubations were analyzed. A total of 368 patients (1.97%) who required chest compressions and/or initiation of the ACLS (advanced cardiac life support) protocol (medication/defibrillation/cardioversion) within 10 minutes (commencement of intubation procedure = time 0) were identified. The patients ranged in age from 17 to 101 years (mean, 68.71 years; median, 67±17.5 years); the male:female ratio was 52 to 48. There was no statistical difference between the non-CA and CA groups regarding race or ethnicity distribution. Each age group (by decades, ie, 30-39 years) was represented consistentwith the age distribution of the overall group. Twenty-one percent of CA patients (n=67) were under 50 years of age, and 46.5% (n=152) were over 70 years of age.
Location of the CA
The number of CAs varied widely by the patient’s location:
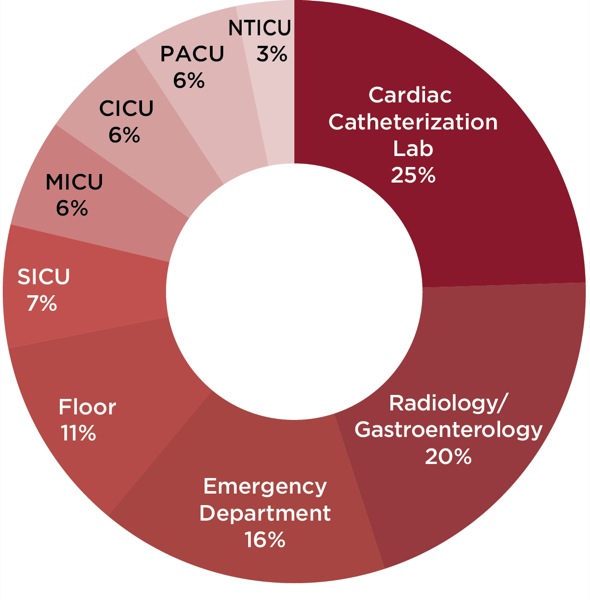
- 85 in hospital wards (23.1% of total);
- 74 in surgical ICUs (20.1%);
- 70 in medical ICUs (19.0%);
- 21 in neurosurgical/trauma ICU (5.7%);
- 22 in coronary care ICU (6.0%);
- 42 in the ED (11.4%);
- 23 in the cardiac catheterization lab (6.3%);
- 25 in the endoscopy/radiology suite (6.8%); and
- 6 in the PACU (1.6%; Table 2).
| Table 2. Cardiac Arrests by Location | ||||||||||
| Floor | MICU | NTICU | ED | SICU | CICU | GI/Rad | PACU | Cath Lab | Total | |
|---|---|---|---|---|---|---|---|---|---|---|
| Intubations | 3,111 | 4,752 | 2,520 | 1,044 | 4,662 | 1,434 | 494 | 408 | 377 | 18,234 |
| Cardiac arrests | 85 | 70 | 21 | 42 | 74 | 22 | 25 | 6 | 23 | 368 |
| Cardiac arrests at location, % | 2.7 | 1.5 | 0.8 | 4.0 | 1.7 | 1.5 | 5.1 | 1.5 | 6.1 | 1.97 |
| Cath Lab, cardiac catheterization lab; CICU, coronary ICU; ED, emergency department; GI/Rad, endoscopy suite/radiology suite; MICU, medical ICU; NTICU, neuro-trauma ICU; SICU, surgical ICU | ||||||||||
Compared with non-CA groups and their locations, the gastrointestinal endoscopy/radiology suite (5.1%) and cardiac catheterization lab (6.1%) were more likely to house CA-NOR patients (Figure 1). The ED (4.0%) and patient ward/floor (2.7%) were both well above the overall average of 1.97% for the entire database (Table 2). Conversely, the neurosurgical/trauma ICU was the least likely location for CA-NOR (0.8%). The surgical ICU rate was 1.7%, and the coronary ICU, medical ICU, and PACU were less likely to have CA-NOR (each at 1.5%; Figure 1).
Comorbidity Burden and Preexisting Disease
The presence or absence of 6 generalized medical diagnoses was documented from the patient’s medical record via their problem list and medical history (Table 3). The analysis of the comorbidity burden suggests that preexisting medical illness was widespread in the vast majority of patients requiring emergency tracheal intubation. The CA-NOR group displayed an increased presence of preexisting disease in 4 of 6 categories that reached statistical significance (Table 4). Those patients with no categorized burden (healthy) were more likely to be in the non-CA group (13.3% vs 5.0%), and those with all 6 categories of pre-existing disease were more likely to be in the CA-NOR group (4.5% vs 10.6%; P<0.01). Those patients with 1 to 5 categorical diseases showed little difference in those suffering a CA and those who did not.
| Table 3. Preexisting Comorbidities |
| Chronic obstructive pulmonary disease |
| Congestive heart failure |
| Coronary artery disease |
| Diabetes mellitus (oral hypoglycemic or insulin dependent) |
| Hypertension |
| Renal insufficiency (serum creatinine >1.6 mg/dL) |
| Table 4. Preexisting Comorbidities Burden | |||
| Cardiac Arrest, % | Database, % | P Value | |
|---|---|---|---|
| Chronic obstructive pulmonary disease | 40.7 | 41.4 | NS |
| Congestive heart failure | 37.3 | 30.2 | 0.005 |
| Coronary artery disease | 48.6 | 39.7 | 0.001 |
| Diabetes mellitus (oral hypoglycemic or insulin dependent) | 44.0 | 40.0 | NS |
| Hypertension | 87.8 | 82.4 | 0.01 |
| Renal insufficiency (serum creatinine >1.6 mg/dL) | 29.6 | 24.0 | 0.02 |
| NS, not significant | |||
Indication for Emergency Airway Management
The medical profiles varied widely. Each critically ill patient typically had more than 1 indication for tracheal intubation. They often had a multitude of medical and surgical issues that accumulated and led to the need for airway intervention (Table 5). The diagnosis documented to be the primary culprit in prompting airway intervention was based on the primary care teams’ documentation in the medical record. The vast majority of patients in the database were intubated for “respiratory insufficiency/failure” rather than for “pure airway protection” in a greater than 9:1 ratio.
However, to improve our understanding of emergency airway management, complications, and any relationship to various disease processes, a more detailed etiology for the deterioration was pursued. Unfortunately, most patients were burdened with multiple maladies contributing to their extreme acuity. Medical documentation was used to determine the primary reason for securing the airway. For example, a morbidly obese patient with acute hypoxic/hypercarbic respiratory failure precipitated by congestive heart failure and pneumonia, acute renal failure with volume overload, metabolic acidosis, and a suspected cerebrovascular accident (categorized as pneumonia as the primary etiology) demonstrates acute critical illness and physiologic turmoil. Conversely, a patient with a drug-induced altered mental status in need of airway protection for support during radiographic head imaging would clearly be categorized as “airway protection.”
Patients were in various phases of resuscitation for their underlying afflictions, placing them at risk for the administration of induction medication or the application of positive pressure ventilation and instituting effective ventilation in the presence of hypercarbia. There was layeringof multiple risk factors contributing to severe hemodynamic alterations, plus the additional risk for airway-related complications (EI, aspiration, multiple attempts, hypoxemia, failed intubation, difficult bag-mask ventilation). Acute cardiopulmonary pathology, sepsis, and upper gastrointestinal bleeding (UGIB) appear to place patients at particular risk for CA (Table 5).
| Table 5. Primary Indication for Emergency Airway Intervention (N=368) | |
| Diagnosis | n (%)a |
|---|---|
| AMI with CHF or shock | 81 (3.8) |
| Pneumonia/aspiration | 76 (4.2) |
| Upper GI bleed | 54 (7.6) |
| Sepsis | 50 (3.2) |
| COPD/respiratory failure | 32 (1.8) |
| Miscellaneousb | 31 (7.9) |
| CHF | 26 (2.7) |
| Airway protectionc | 11 (0.9) |
|
a Cardiac arrest rate (%) by the primary pathologic condition prompting intubation.
b Includes pulmonary embolus, cardiac tamponade, pneumothorax/tension pneumothorax.
c Includes altered mental status, alcohol withdrawal, drug overdose, etc.
AMI, acute myocardial infarction; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; GI,gastrointestinal
|
|
Conversely, CA was underrepresented in the trauma (1.1%), neurologic (1.0%), and neurosurgical populations (intracerebral bleed, 0.7%) and in those being intubated solely for airway protection (0.9%) for an underlying issue: alcohol withdrawal, drug overdose, encephalopathy, metabolic disorders, and seizures. Specifically, acute myocardial injury complicated by congestive heart failure or cardiogenic shock (4.1%) and aspiration pneumonia (5.0%) increased the odds ratio that CA would complicate the intubation process. Patients in the throes of cardiac dysrhythmia disturbances, too, are at increased risk for post-intubation CA (5.5%).
Pneumonia alone (2.0%) did not appear to put the patient at increased risk compared with the overall database (1.97%), whereas pulmonary or abdominal sepsis more than doubled the CA risk. Although patients with UGIB (7.6%, Table 5) were often intubated to provide airway protection, their pathophysiologic profile was clearly higher risk than other mentioned miscellaneous etiologies for requiring airway protection, as they typically had concomitant hypovolemia, active bleeding, and insufficient resuscitation at the time of intubation. These issues were often coupled with multiple organ dysfunction (eg, ascites, liver failure, renal failure, altered mental status, and coagulopathies) and a markedly increased risk for regurgitation and aspiration. Moreover, positive pressure ventilation may lead to a decline of cardiopulmonary function by altering ventricular input/output. These effects were particularly distressing for those with acute myocardial injury, diminished myocardial function, and cardiogenic shock.
Moreover, maladies in the miscellaneous category represent acute cardiopulmonary stressors that have significant implications for anesthesia induction and maintenance in the OR. Pulmonary embolus of any etiology (thrombus, fat, amniotic, air, glue, white cell demargination, etc), cardiac tamponade, and pneumothorax/tension pneumothorax elevate the risk to an unprecedented level (7.9%). For example, pericardial tamponade restricts diastolic filling. Adequate intravascular volume and preload are required to augment end-diastolic volume and stroke volume. In the face of compromised stroke volume, the heart relies on maintenance of contractility, an increased heart rate, and vasoconstriction to maintain blood pressure. Even reduced anesthetic dosing and the application of positive pressure ventilation may alter the patient’s heart rate, preload, afterload, or contractility, leading to CVC.
Their presentation under emergent and dire circumstances therefore elevates the risk level prohibitively. The complete description of the altered physiologic status and nature of the patient’s acute physiologic deterioration is, unfortunately, absent when a single disease entity is assigned as a primary causative factor for airway intervention. Various body systems interact, complement, and detract from each other in a multitude of pathways. Branding patients into a single disease category serves as a means to distinguish them and may assist practitioners to improve our risk assessment for emergency airway intervention outside the OR. Labeling 90% as “respiratory insufficiency” and 10% as “altered mental status/airway protection” would not allow any distinction between UGIB patients with multiple organ dysfunction needing airway protection versus a young, previously healthy adult who is now experiencing alcohol-induced seizures. It was uncommon for a patient to have only a single disease process or single-organ compromise culminating in tracheal intubation.
Method of Patient Preparation
The preparation of the patient for an emergency intubation outside the OR varies by location, available medication, the patient’s critical care state, hemodynamic instability, and comorbidities, coupled with the preferences and experiences of the airway team. Hemodynamic stability may be short-lived after the administration of induction agents, regardless of reduced dosing, compared with the more stable OR milieu. Indeed, a stable bedside hemodynamic profile may deceive the airway team into a false sense of security, leading to more than is actually needed or will be tolerated. Further, the patient’s mental status and level of consciousness will certainly affect the team’s decision about preparation for intubation.
Certainly, known or suspected difficult airway status may be gleaned by the medical record, helpful staff, or a rapid bedside assessment for the presence of difficult airway characteristics. Many practitioners have developed their own recipes to prepare critically ill patients for intubation. Flexibility is a prudent option given the varied presentation of an acutely ill patient in need of airway control. Some patients will not require any medication, due to a depressed mental status, but may need the medication only to tame the stimulatory effects of laryngoscopy and the intubation itself. Finally, topical local anesthesia (TLA) is an option for patients with and without difficult airway characteristics to reduce or eliminate the need for IV agents that may generate significant hemodynamic alterations.
Parenteral medication was administered in limited, incremental doses in 78% of patients who suffered a CA. A majority received neuromuscular blocking agents (NMBAs; 62%). The remaining patients received only topical anesthesia of the airway, or no medication was administered to prepare for the airway procedure (Table 6). Medications included midazolam (0.02-0.04 mg/kg; 2-3.5 mg), morphine (0.03-0.04 mg/kg; 2-3 mg; Group 1 only), diazepam (0.12 mg/kg; 10 mg; Group 1 only), propofol (0.8-1.5 mg/kg; 100-225 mg), and etomidate (0.12-0.22 mg/kg; 9-24 mg).
| Table 6. Patient Preparation for Tracheal Intubation (N=368) | ||||||||
| No Sedative-Hypnotics | Topical Only | MS | Barb | Benzo | Etomidate | Propofol | Total | |
|---|---|---|---|---|---|---|---|---|
| Patients, n | 49 | 27 | 3 | 6 | 56 | 143 | 84 | 368 |
| Non-Dep | 2 | 2 | 2 | 3 | 14 | 18 | 16 | 57 |
| Dep | 9 | 11 | 1 | 1 | 43 | 73 | 34 | 172 |
| Barb, methohexital, thiopental; Benzo, midazolam, diazepam, lorazepam; Dep, depolarizing agent (succinylcholine); MS, morphine, fentanyl; Non-Dep, non-depolarizing agent (pancuronium/vecuronium/rocuronium); topical only,topical/local anesthesia | ||||||||
The 228 cases who received NMBAs were administered a depolarizing agent (succinylcholine, 172 cases; 75%) or a non-depolarizing agent (pancuronium, vecuronium, or rocuronium, 57 cases; 25%), compared with the non-CA group (39.8% received NMBAs; Table 6). Both CA and non-CA exposure to NMBAs was in an approximate 80:20 ratio of depolarizing agent to non-depolarizing agent. There were no cases involving hyperkalemia related to succinylcholine administration leading to CA. NMBAs were used in 35 cases without any sedative-hypnotics (n=12) or after administration of TL A (n=23) based on depressed mental status (unconscious) or prior administration of sedative-hypnotics/analgesics by the primary care team before the arrival of the anesthesia team at the bedside (eg, benzodiazepine administration for delirium tremens, seizures, or agitation).
Presenting Cardiac Rhythm With CVC
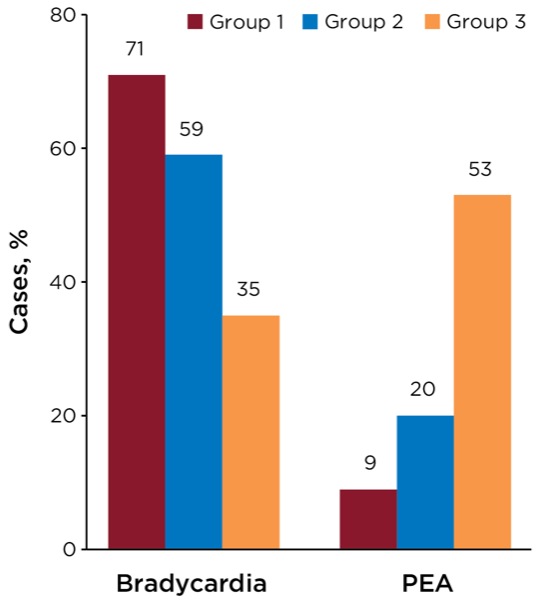
The primary documented rhythm disturbance that led to CVC, with subsequent initiation of ACLS-based resuscitative measures, appeared to fall into 3 separate categories. The most common presenting rhythm disturbance was a progressive decline in heart rate to the range of 20 to 35 beats per minute, which was noted in 178 (48.4%) of the 368 CAs. Nearly one-half of the bradycardia cases (49.4%; n=88) progressed to asystole. Ninety-six patients (26.3%) developed CA (loss of palpable pulses or arterial line tracing) while maintaining a heart rate (pulseless electrical activity [PEA]).
The remaining patients developed ventricular tachycardia/fibrillation (VT/VF) during or immediately after the airway management procedure. The preponderance of bradycardic arrest victims (69.2%) experienced some degree of oxygen desaturation, via pulse oximetry. Maintenance of oxygen saturation (SpO2) greater than 90% occurred in 30.8% of these cases, while 10% experienced mild hypoxemia (SpO2 <90%) and 59% experienced severe hypoxemia (SpO2 <80%). By contrast, desaturation was less common in both cases of PEA CA (30.5%) and ventricular-based dysrhythmia (33.3%).
Of note, the character of the CA rhythm changed over the three time periods. This likely reflects the reduction of hypoxia-driven declines in heart rate, thus a reduction in the incidence of bradycardic CA. There was a stepwise reduction in the rate of airway-related complications over the 3 groups, as reflected in Figure 2, which illustrates the decline in bradycardia CA and the increased prevalence of PEA as the advancement in airway management schema improved over time.
A Subcategory of Post-Intubation CA
One of every 10 CA cases arose from post-intubation difficulty with ventilation and oxygen delivery (luminal or tracheobronchial tree obstruction) via the newly placed ETT. This took place regardless of whether the intubation itself was straightforward or difficult. This subcategory of “obstructed CA” was due to an acute accumulation of blood, thickened secretions, particulate matter, or gastric content that contributed to obstruction of the ETT lumen and/or tracheobronchial tree (n=37). This subgroup experienced a 100% incidence of desaturation.
The anesthesia airway team encountered high resistance during attempts to deliver positive pressure ventilation after intubation, thus leading to hypoxemia and cardiac dysrhythmia with subsequent CA. Despite vigorous saline irrigation and therapeutic bronchoscopy in many of the cases, nearly 80% prompted the removal and replacement of the ETT.
| Table 7. Airway-Related Complications: Cardiac Arrest Categories Versus Non–Cardiac Arrest Patients | ||||||
| Frequency, n (%) | Multi Att (3+), % | Hypoxemia SpO2 <90%, % | EI, % | Regurgitation, % | Aspiration, % | |
|---|---|---|---|---|---|---|
| Bradycardic arrest | 199 (54.1) | 52.2 | 72.8 | 51.7 | 26.7 | 13.9 |
| Nonobstructed bradycardia | 178 (48.4) | 59.6 | 68 | 53.4 | 27.0 | 14.1 |
| PEA with heart rate | 96 (26.1) | 13.5 | 30.5 | 10.4 | 6.3 | 4.2 |
| VT/VF | 73 (19.8) | 28.8 | 47.9 | 19.2 | 19.1 | 12.3 |
| Nonobstructed VT/VF | 57 (15.5) | 36.8 | 33.3 | 21.5 | 24.6 | 15.8 |
| Obstructed bradycardia | 21 (5.7) | 33.3 | 100 | 38.1 | 4.8 | 4.8 |
| Obstructed VT/VF | 16 (4.3) | 0 | 100 | 12.5 | 0 | 0 |
| Total cardiac arrest cases, % | 368 (100) | 35.5 | 56.3 | 34.6 | 20.5 | 11.3 |
| Non-cardiac arrest database, % | 18,267 (100) | 9.0 | 14.9 | 6.0 | 1.1 | 0.4 |
| EI, esophageal intubation; Multi Att, 3+ attempts; PEA, pulseless electrical activity; SpO2, oxygen saturation; VT/VF,ventricular tachycardia/fibrillation | ||||||
The overall group of CA patients experienced a considerably higher incidence of airway-related complications compared with non-CA patients (Table 7). Setting aside those patients who suffered obstruction CA, those who suffered a bradycardic arrest experienced a considerably higher incidence of airway-related complications compared with patients who experienced either nonobstructive PEA or VT/VF CA. The impact of airway-related complications on the incidence of CA in the database was significant (Table 7). However, when the CA incidence is categorized by time period, there was a vast reduction over time of the impact that airway-related complications had on CA as the primary causative factor during emergency intubation (Table 8).
| Table 8. Cardiac Arrest by Time Period: Airway-Related Complications (%) | ||||||
| 3+ Attempts | SpO2 <90% | EI | Regurgitation | Aspiration | Cric | |
|---|---|---|---|---|---|---|
| Group 1 (123; 3.7%) | 70.7 | 76.4 | 61.8 | 30.0 | 17.1 | 18.7 |
| Group 2 (103; 2.1%) | 35.0 | 56.3 | 23.3 | 24.3 | 10.7 | 5.8 |
| Group 3 (142; 1.4%) | 16.9 | 38.0 | 14.1 | 7.0 | 2.8 | 2.8 |
| Non-cardiac arrest (n=18,267) | 8.9 | 14.9 | 6.9 | 1.1 | 0.4 | 0.6 |
| Group 1 (1990 to late 1995); Group 2 (late 1995 to mid-2006); Group 3 (mid-2006 to 2017). Cric, cricothyrotomy performed; EI, esophageal intubation; SpO2, oxygen saturation | ||||||
Characteristics of Airway Management Complications
A variety of airway complications were prevalent in the CA group. The consequence of each complication, in many cases, was its effect on the incidence of hypoxemia. The incidence of hypoxemia during and immediately after the intubation procedure (56.3%) was highlighted by the 100% rate of desaturation in the obstructive CA group. Tracheal intubation was successful in 94.2% of cases, with the remaining 19 cases requiring a surgical airway. Two patients in Group 1 died without establishing an airway (massive head and neck engorgement, a short neck, and marked obesity).
Tracheal Intubation Attempts
First-pass intubation success varied widely in the CA groups, with only 18% success (Group 1 vs 60.3% in non-CA Group 1) rising to 52% (Group 2 vs 65.5% in non-CA Group 2) and peaking at 63.2% (Group 3 vs 76.6% in non-CA Group 3). Instances of multiple intubation attempts (3+) in the overall database were 1 in 11 cases (8.8%) compared with 39.9% of cases in the CA group. Before deployment of accessory airway devices (Group 1), the rate of multiple attempts was considerably higher: 70.7% in the CA group versus 16.1% in the non-CA group (Table 8). The immediate availability of accessory devices (Group 2) afforded nearly a 50% reduction in multiple attempts (3+) compared with Group 1 (70.7%→35%; Table 8).
The addition of VAL technology (Group 3) provided a further 50% reduction in multiple attempts in the CA group (16.7%). Similar reductions in the rate of multiple attempts were noted in non-CA cases over the 3 time periods (16.1%→8.3%→6.7%; Table 8). The incidence of a surgical airway (“cric”) in the CA group fell from 18.7% (23 cases) to 5.8% (6 cases) in Group 2 to 2.8% (4 cases) in Group 3 (Table 8). The immediate availability of accessory airway devices, particularly video laryngoscopes, assisted the airway team in reducing overall intubation attempts and the need for surgical airway rescue. This reduction was reflected in the non-CA group over the 3 time periods (0.9%→0.6%→0.2%).
Esophageal Intubation
The rate of EI for the entire database was 6.9%, with a stepwise reduction observed over the 3 time periods: 16.7% in Group 1 versus 10.4% in Group 2 versus 2.6% in Group 3 (Table 8). The impressive 38% reduction from Group 1 to 2 was overshadowed by the 75% reduction from Group 2 to 3; the overall reduction from Group 1 to 3 was 84%. The incidence of EI in the entire CA group was nearly five times higher (34.6%) than that of the entire non-CA group (6.9%) (Tables 7 and 8). Although more than one-half of EIs were a single event, nearly as many patients suffered multiple esophageal misplacements.
A total of 210 ETT misplacements were recorded in 127 CA patients, of which 58 experienced more than 1 EI (69 patients, 1 EI; 42 patients, 2 EIs; 9 patients, 3 EIs; 5 patients, 4 EIs; 2 patients, 5 EIs), mostly during the first 2 periods. Only 10 CA patients of these 127 (7.9%) maintained their SpO2 greater than 90% in the face of ETT misplacement. This far outdistanced the non-CA group, who suffered EI and did not desaturate less than 90% (489/1,322; 38.5%; P<0.001). Additional airway-related complications associated with EI were regurgitation and aspiration. The CA patients who experienced EI had an increased incidence of regurgitation (46.0%) and aspiration (24.8%) over the non-CA group with EI (8.9% and 3.8%, respectively; P<0.001).
Analyzing the airway complications from a different perspective demonstrated the impact that EI had on patient safety. Those who experienced any degree of oxygen desaturation (<90%) during the intubation procedure were more likely to have suffered EI (57.9% CA vs 24.8% non-CA groups; P<0.001). Likewise, regurgitating gastric contents during the intubation procedure was more likely in patients who experienced EI (77.6% CA vs 45.6% non-CA; P<0.001). Moreover, aspiration of gastric contents related to EI was more common in CA versus non-CA patients (75.7% vs 51.4%; P<0.02). The rate of EI was notably higher in bradycardic CA (53.4%) compared with nonobstructed VT/VF CA (21.5%) and PEA CA (10.4%), which is consistent with the much higher rate of oxygen desaturation likely contributing to bradycardic CA cases (Table 7). Such occurrences contributed to hypoxemia, multiple intubation attempts, and difficult airway management.
The 3 groups differed in the overall rate of CA EI (61.8%, Group 1 vs 23.3%, Group 2 vs 14.1%, Group 3; Table 8). This reflects the progressively fewer multiple intubation attempts (3+) and higher first-pass success rates that were likely based on the clinician’s immediate access and utilization of accessory airway devices, particularly VAL. However, the two more recent time periods showed a reduction in the rate of multiple (repeat) EI compared with the earlier period:
- Group 1: 43 of 76 (56.6% vs non-CA: 21.6%; P<0.001);
- Group 2: 12 of 24 (50% vs non-CA: 11.8%; P<0.001); and
- Group 3: 3 of 27 (11.1% vs non-CA: 3.8%; P<0.001).
Regurgitation and Aspiration
The incidence of regurgitation (1.6%) and aspiration (0.6%) in the entire emergency intubation database underwent a significant reduction over the 3 time periods (Table 8). Peaking at 2.9% in Group 1, the regurgitation rate was lowered by 65% to 1.0% (in Group 2) with a further 40% decline (0.6%) in the VAL era (Group 3, Table 8). Equally evident was the reduction in aspiration cases, by 66% from 1.5% (Group 1) to 0.5% (Group 2), followed by a 60% drop to 0.2% (Group 3). The overall rate of regurgitation and aspiration of gastric contents for the CA group was 18.8% and 10.6%, respectively, which was magnitudes greater than the non-CA group, at 1.1% and 0.4%. Sixty of 69 regurgitation victims (95.6%) and 36 of 39 aspiration cases (92.3%) had concomitant hypoxemia during the intubation event.
Regurgitation was more likely with multiple EIs (45%) compared with a single EI (33.3%) and those who did not undergo an EI (21.2%). The airway team witnessed newly regurgitated gastric contents below the glottis or physically removed the fresh gastric material by suctioning the tracheal tube in 39 of the 69 regurgitant victims (aspiration, 56.5%). Of note, nonobstructed bradycardic and VT/VF CA had higher rates of regurgitation and aspiration, respectively (27.0% and 14.1%; bradycardic CA) and (24.6% and 15.8%; VT/VF CA), compared with PEA CA (6.3% and 4.2%; Table 7). As previously noted, the impact of immediate availability of accessory airway devices and ETT-verifying equipment was profound in the overall database (~60% from both Group 1→2 and 2→3; Table 8). In the CA group, however, there was only a 20% decline in regurgitation (Group 1→2) coupled with a 71% reduction from Group 2→3. Aspiration cases in the CA group dropped significantly (Group 1→2) and then underwent a further healthy reduction from Group 2→3 (Table 8).
Outcome
Overall, 58.3% died before hospital discharge. Thirty-three patients (10.7%) died within 24 hours, of which 11 died immediately (<60 minutes) after withdrawal of care or lack of response to care. Two of the 11 immediate deaths (both of which occurred in Group 1) died without their airway being secured by any means due to anatomic limitations (profound head/neck/airway edema). A significant number of CA patients survived the event but later succumbed after extended hospital care (2-39 days; 146 patients; 47.6%). Survivors accounted for 41.7% of CA victims. The outcome of 20 of the 368 CA patients was indeterminate.
Discussion
Hemodynamic perturbations with inflated blood pressure ranges, heart rate irregularities, and rate extremes combined with cardiopulmonary compromise, peripheral vasodilation, and volume under- and overloading may be further amplified by parenterally administered sedative-hypnotic/analgesic agents. This data review provides insight into the contributing factors that were associated with CA during emergency airway management outside the OR. The underlying acute pathophysiologic deterioration of the patient, the administration of induction medications, and airway-related critical events all appear to provide the inciting stress that singly or in combination culminated in CA.
Oxygenation and ventilation difficulties within the OR environment have accounted for nearly one-third of all anesthetic deaths.23,24 A critically ill patient who requires emergency tracheal intubation outside the OR is at risk for numerous airway and hemodynamic complications, and remains a formidable challenge for the airway manager. This report provides specific details of the contributing factors for CA during emergency airway management outside the OR and suggests a relatively common rate of occurrence for this catastrophic event (2%). Furthermore, this report lends support for the recommendations of the ASA Task Force on the Management of the Difficult Airway for having immediate access to advanced airway devices and ETT-verifying devices.13
The attention given to the complications of emergency airway management in remote locations in the anesthesia literature is not commensurate to the incidence of these critical events and patient injury. Further, given the grave consequences of airway difficulties and associated hypoxic brain injury, pharyngolaryngeal trauma, and death, all of which have been reported, the attention CA deserves is fortunately increasing but remains in its infancy. The relationship between CA and emergency airway management and intubation in the ED and other non-OR areas was suggested by a variety of investigators.7-12
As previously stated, CA in the OR is relatively rare (0.05%-0.02%), but this critical event is associated with several underlying factors: the elderly, ASA physical status III and IV, pediatrics, major surgical procedures, and emergency surgery.1-5,23,24 Anesthesia-related difficulty with oxygenation and ventilation account for only a small proportion of this total.2,3,23,24 The intraoperative use of oximetry, capnography, and ventilator disconnect monitors, plus a major educational effort by the ASA, have all been credited for the decreasing rate of respiratory-based CAs, which now stands at less than 1 case per 10,000 anesthetics.2,3,23-26 Despite the significant intrinsic differences between the reported OR rate and the non-OR patient population in this database, CA during emergency intubation in a remote location (1 arrest for every 50 intubations, overall) is sharply higher than its intraoperative counterpart. However, the incorporation of the suggestions of having immediate access to accessory airway devices and using ETT-verifying devices, such as capnography or a bulb syringe technique, as posed by the ASA Task Force on the Management of the Difficult Airway, appeared to reduce the overall rate of CAs, especially those whose etiology is hypoxemia.8,13
Several options are available for preparation of the patient, depending on the clinical reason prompting intubation, current and past medical conditions, the airway manager’s experience, and judgment of the patient’s presumed needs. Although each patient is critically ill, there is a gradation of preexisting and current pathology, and many practitioners tailor an induction regimen specifically to a patient’s needs. The literature has offered several protocols for induction and intubation in the ER, often ignoring the individual’s clinical illness and simply grouping the patients by induction technique.27-33
Sedative-hypnotic and opioid medications may blunt airway reflexes, induce apnea, alter hemodynamics, and worsen an already tenuous airway. Although it is difficult to specifically document the exact effect of parenterally administered sedatives in this group of CA patients, the anesthesia preparation contributed to hemodynamic instability to varying degrees in many of the patients, despite their having received incremental doses of sedatives and/or opioids in proportionately lower dosages than is typical in an elective OR situation. However, anesthetic medications remain an important piece of a multifactorial puzzle, underlying the etiology for CA in the remote location.
Anesthetic preparation likely served as the primary factor contributing to hemodynamic collapse in several patients who suffered CA-NOR without any intervening airway complications. Commonly administered dosing of induction agents in the elective OR setting (ie, 1.3-1.8 mg/kg propofol and 0.25-0.3 mg/kg etomidate) likely contributed to increasing hemodynamic instability in critically ill patients undergoing emergent intubation.
NMBAs were used relatively sparingly in the overall database (39%), reflecting the practice of maintaining spontaneous ventilation for emergency tracheal intubation. By contrast, 62% of the CA victims received an NMBA during the intubation procedure. The safety of using an NMBA during emergency intubation outside the OR and by nonanesthesia personnel has been questioned, and many potential hazards probably remain underreported. NMBAs appear to be a reasonably safe adjunct for emergency intubation when managed by experienced personnel whose rationale for their use is tempered by an assessment of the appropriateness for their administration, coupled with an evaluation of the patient’s health and airway status.27-33Nonetheless, NMBAs are administered frequently, especially for an emergency airway, by nonanesthesiologists.
It is the author’s opinion that a prudent approach for the use of NMBAs in a remote location would be based on the patient’s clinical circumstance and the airway manager’s experience, rather than a prearranged, universal recipe or protocol. More imperatively, their use should be pursued only if alternative methods of ventilating and securing the airway are immediately available should conventional intubation methods fail.
Factors Contributing to CA
The etiology of CA is typically multifactorial. On the basis of the findings from this database, several clinical factors appear to contribute to this catastrophe. The emergent nature of the airway intervention, the clinical disease state that prompted intubation, the hemodynamic impact from anesthetic medications, the initiation of positive pressure ventilation, and any intervening airway-related complication that contributes to profound hypoxemia may all contribute (Table 9).1-5,8,12 The underlying pathophysiology appears to be a major contributor to the risk for CA, as is suggested in the group whose CA occurred after a relatively uneventful intubation. A relative anesthetic overdose can lead to cardiac collapse, especially in the presence of cardiopulmonary conditions such as shock, major cardiac injury and dysfunction, systemic vasodilation (sepsis), tension pneumothorax, pulmonary embolism, severe hypovolemia of any etiology, and cardiac tamponade after tracheal intubation. Individual sensitivity to the application of positive end-expiratory pressure (PEEP) ventilation and the possible development of “auto-PEEP” can dramatically alter venous return, afterload, and systemic blood pressure.34 Furthermore, overzealous ventilation leading to a relative or absolute hypocarbic state in the previously hypercarbic patient may further erode sympathetic tone (Table 9).4-6,8,23,24,34
| Table 9. Factors Contributing to Cardiac Arrest3,5-8,10,23,24 |
| Anesthetic medications |
| Auto or intrinsic PEEP |
| Decrease in patient work |
| Excessive parasympathetic tone |
| Hyperkalemia (succinylcholine) |
| Hyperventilation with preexisting hypercarbia |
| Hypoxia-related hemodynamic deterioration |
| Loss of spontaneous respirations |
| Mediastinal pathology |
| PEEP |
| Positive pressure ventilation |
| Preload-dependent physiology, valvular heart disease, congestive heart failure, pulmonary embolus, right ventricular failure, restrictive pericarditis, cardiac tamponade, pneumothorax, tension pneumothorax |
| Sympathetic surge, vasovagal response |
| Underlying disease process (ie, myocardial insufficiency) |
| Volume imbalances (sepsis, diuretics, hemorrhage) |
| PEEP, positive end-expiratory pressure |
Airway management difficulties, singly or in combination with other factors, added further to the rapid hemodynamic collapse in the majority of cases (Table 9). In total, these factors appear to escalate the rate of CA during emergency tracheal intubation outside the OR.7-12 Succinylcholine-induced hyperkalemia, as has been noted in the pediatric population, did not appear to be a factor in this collection of adult data.
Severe bradycardia and sinus arrest are well known yet uncommon complications of the stress from laryngoscopy and intubation.34,35 A sympathetic surge during airway manipulation may potentiate alterations in ventricular compliance and contractility by increased right and left afterload. Moreover, an unblunted sympathetic surge mediated through ventricular C fibers may induce a vasovagal response similar to a neurally mediated syncopal episode. CA in the face of a seemingly uneventful intubation may, on occasion, be provoked by the patient’s own parasympathetic influences from a single vigorous laryngoscopy or from current medication regimens (ie, beta-blockers). In addition, the author has observed many exhausted, air-hungry, critically ill patients who require tracheal intubation, requiring little or no sedation, who develop a near-immediate relaxed, sleeplike state, followed by significant hypotension.
A rapid but progressive slowing of the pulse rate that culminated in bradycardia (<30 beats per minute) or asystole with an unobtainable blood pressure was the common downward spiral leading to instituting CPR in 54.1% of the CAs over the 28-year period. Although a progressive bradycardia in this clinical scenario is not-a new observation,3-5 it reinforces that such a rhythm is often the last step before external cardiac massage. This finding is juxtaposed to the reported primary CA rhythm in 4 recent reports of CA-NOR, which found PEA to be the overwhelming rhythm.36-39 Moreover, airway-related complications associated with hypoxia were uncommonly mentioned as an etiology factor in CA cases.9,36-38
Conversely, this vast database, covering three decades compared with a 1- to 2-year period, displayed a transition away from airway-related complications as the primary etiological factor. The majority of CA-NOR cases were based on a multitude of factors: primarily, the acute clinical deterioration that prompted tracheal intubation and, commonly, a hypoxic insult, particularly in the earlier time periods. In Group 3 (2006-2017), bradycardia accounted for only one-fourth of the instances of CA, while PEA rhythm was the most common. The combination of the physiologic alterations with the institution of positive pressure ventilation, particularly if post-intubation hyperventilation was practiced, may lead to further cardiopulmonary deterioration.
The clinician has little control over the physical status of the patient presenting for emergent airway management, but we can have a tremendous impact on patient safety if we can offer improved airway management strategies to minimize any further physiologic insult to an already “crashing” patient.39,40 The abandonment of conventional intubation measures and a rapid move to a rescue option, coupled with the use of ETT-verifying devices, appear to lower the rate of CA when airway mishaps play a prominent role in the CA’s etiology.
Underreporting of CA Rates
The number of CAs may be higher than actually reported in this series, based on the fact that a questionnaire had to be completed for entry into the database during the majority of the data collection time frame. Despite attempts by the author to verify the accuracy of the recorded information via the post-procedure questionnaire, a major concern with any spontaneous reporting system is underreporting.41,42 Self-reporting of adverse events is often limited by the recognition of the adverse event, the care provider’s tendency to minimize the importance of such an event, or the fear of any consequences that may result from the reporting of one’s own complications.42-44 The 2% CA rate is at best a rough guess, but based on reporting tendencies, the underreporting of this critical event is probably the rule rather than the exception.
Recent publications in the emergency medicine literature reflect similar findings about post-intubation CA rates. Markedly higher rates of CA are reported (4.0% and up to nearly 20% in selected groups of patients), but the reporting criteria differ from the present database. We report CA that had occurred during or within 10 minutes of the commencement of the intubation procedure (time 0). Some report much higher rates but include a longer time frame (eg, 20 minutes), which may capture a significantly higher number of cases of CA after intubation. Likewise, we reported all CAs across the entire database, whereas others who reported higher CA rates did so in select patient groups with higher risks for CA: preintubation hemodynamic instability, namely, hypotension, shock, or an elevated shock index.9,36-38 It would appear that there is much agreement that intubation can lead to significant hypotension at a frequent rate, and both pre- and post-intubation hypotension seem to be markers for CA. Also, of note, is the induction regimen used in the vast majority of intubations, particularly in the ED setting: sedative-hypnotics with paralysis appear as a routine induction method.9,36-38 Again, patient preparation customized to the patient’s status rather than standard recipe, may be an alternative worse reviewing.
Our database was uncontrolled, as the airway team provided medications according to their own experience and judgment of the patients’ needs and clinical condition(s). This, of course, may affect the rate of CA-NOR. Although certainly not applicable to all patients in extremis, it may be warranted to examine the role of patient preparation using TLA alone or combined with markedly reduced dosing of sedative-hypnotics, narcotics, and/or anxiolytics as an alternative to more standard induction/paralysis for intubation. Reviewing a subgroup of patients from our database (preintubation cardiogenic shock, n=1,910), those patients undergoing TLA preparation (n=264) did not experience CA versus a 4.7% rate for the remaining cardiogenic shock patients receiving induction agents, with or without paralysis. Other high-risk clinical conditions prompting the need for NOR intubation that appear to benefit from TLA preparation with a reduced incidence of CA-NOR include:
- acute myocardial infarction (2.5% vs 3.2%-non-TLA);
- miscellaneous cardiopulmonary maladies (tamponade, embolus, zero cases vs 7.9%);
- cardiac dysrhythmias (1.0% vs 5.5%);
- aspiration (2.6% vs 5.0%);
- pneumonia (1.2% vs 2.0%); and
- abdominal sepsis (zero cases vs 3.2%).
Moreover, the CA rate in those with preintubation hypotension (mean arterial pressure <60 mm Hg) undergoing TLA preparation was only slightly elevated (2.7%) over the entire database incidence of CA (1.97%). Further research is needed to clarify this point. Is treatment with preinduction fluid resuscitation and/or vasopressors to a specific end point possibly helpful to decrease the rate of post-intubation CA in these selected patient groups?45 Jaber et al instituted an intubation bundle checklist in the ICU.45 Their intervention list included providing a fluid bolus if indicated to treat preintubation hypotension. They showed a significantly lower rate of mild to moderate and life-threatening complications. Their efforts also found a lower rate of post-intubation CA, although this did not reach statistical significance.45
Nevertheless, preexisting hypotension, shock, vasodilation, or an altered shock index in the preintubation phase are cause for concern. Many preexisting morbidities and current systemic conditions place any patient at grave risk for CVC and hemodynamic pertubations.6 These factors, together with an intubation procedure coupled with post-intubation positive pressure ventilation, induction agents, and preexisting cardiopulmonary compromise and other metabolic/acid–base disorders, means the airway team has a considerable challenge at hand and a potential catastrophe.8,9,36-38
Conclusion
The etiology of CA during emergency intubation outside the OR is most often multifactorial, with a wide array of clinical conditions that contribute to profound hemodynamic deterioration, which are often combined with or compounded by airway-related mishaps. Succinylcholine-induced hyperkalemia was not found to be a contributing factor for CA in this adult population. This database reviewed the evidence that the ASA’s “Practice Guidelines for Management of the Difficult Airway” had a demonstrable effect on the rate of CA-NOR by offering immediate access to ETT-verifying devices and advanced airway devices.13 Furthermore, the addition of VAL to the airway teams’ armamentarium appears to have lowered the risk for airway difficulties that may be a prominent etiological factor of CA-NOR.
Overall, the incidence of CA-NOR has fallen considerably over the three time periods.. Moreover, the characteristic CA rhythm presented to the airway team has shifted from hypoxia-induced bradycardia to PEA, and the incidence of hypoxia related to airway complications has fallen with the deployment of advanced airway adjuncts and universal EtCO2 monitoring during the intubation procedure.39,40 Airway management difficulties leading to CA appear to be lowered further (Group 2→Group 3) by the bedside deployment of adjuncts and EtCO2 capabilities coupled with the ubiquitous availability and use of VAL instrumentation. In those clinical conditions that pose particularly high risk for CA, it appears that TLA preparation for the airway manipulation may lower the risk of complete cardiovascular collapse and cardiac arrest.
Therefore, ensuring immediate access to advanced airway rescue adjuncts, including VAL and ETT-verifying devices, appears warranted. Expending the time, effort, and cost to ensure the immediate availability of advanced airway rescue options and ETT-verifying devices in remote locations, coupled with the knowledge of how and when to use them properly, is reasonable and justified, and should be considered the standard of care for emergency airway management outside the OR.
References
- Nunnally ME, O’Connor MF, Kordylewski H, et al. The incidence and risk factors for perioperative cardiac arrest observed in the national anesthesia clinical outcomes registry. Anesth Analg. 2015;120(2):364-370.
- Olsson GL, Hallen B. Cardiac arrest during anaesthesia. A computer-aided study in 250,543 anaesthetics. Acta Anaesthesiol Scand. 1988;32(8):653-664.
- Keenan RL, Boyan CP. Decreasing frequency of anesthetic cardiac arrests. J Clin Anesth. 1991;3(5):354-357.
- Newland MC, Ellis SJ, Lydiatt CA, et al. Anesthetic-related cardiac arrest and its mortality: a report covering 72,959 anesthetics over 10 years from a US teaching hospital. Anesthesiology. 2002;97(1):108-115.
- Sprung J, Warner ME, Contreras MG, et al. Predictors of survival following cardiac arrest in patients undergoing noncardiac surgery: a study of 518,294 patients at a tertiary referral center. Anesthesiology. 2003;99(2):259-269.
- Perbet S, De Jong A, Delmas J, et al. Incidence of and risk factors for severe cardiovascular collapse after endotracheal intubation in the ICU: a multicenter observational study. Crit Care. 2015;19(1):257.
- Schwartz DE, Matthay MA, Cohen NH. Death and other complications of emergency airway management in critically ill adults. A prospective investigation of 297 tracheal intubations. Anesthesiology. 1995;82(2):367-376.
- Mort TC. The incidence and risk factors for cardiac arrest during emergency tracheal intubation: a justification for incorporating the ASA Guidelines in the remote location. J Clin Anesth. 2004;16(7):508-516.
- Heffner AC, Swords DS, Neale MN, et al. Incidence and factors associated with cardiac arrest complicating emergency airway management. Resuscitation. 2013;84(11):1500-1504.
- Smischney NJ, Seisa MO, Heise KJ, et al. Predictors of hemodynamic derangement during intubation in the critically ill: a nested case-control study of hemodynamic management-Part II. J Crit Care. 2018;44(4):179-184.
- Wardi G, Villar J, Nguyen T, et al. Factors and outcomes associated with inpatient cardiac arrest following emergent endotracheal intubation. Resuscitation. 2017;121(12):76-80.
- Schwab TM, Greaves TH. Cardiac arrest as a possible sequela of critical airway management and intubation. Am J Emerg Med. 1998;16(6):609-612.
- Apfelbaum JL, Hagberg CA, Caplan RA, et al. Practice guidelines for management of the difficult airway: an updated report by the American Society of Anesthesiologists Task Force on Management of the Difficult Airway. Anesthesiology. 2013;118(2):251-270.
- Arulkumaran N, Lowe J, Ions R, et al. Videolaryngoscopy versus direct laryngoscopy for emergency orotracheal intubation outside the operating room: a systematic review and meta-analysis. Br J Anaesth. 2018;120(4):712-724.
- Mosier JM, Whitmore SP, Bloom JW, et al. Video laryngoscopy improves intubation success and reduces esophageal intubations compared to direct laryngoscopy in the medical intensive care unit. Crit Care. 2013;17(5):R237.
- Kory P, Guevarra K, Mathew JP, et al. The impact of video laryngoscopy use during urgent endotracheal intubation in the critically ill. Anesth Analg. 2013;117(1):144-149.
- Griesdale DE, Liu D, McKinney J, et al. Glidescope® video-laryngoscopy versus direct laryngoscopy for endotracheal intubation: a systematic review and meta-analysis. Can J Anaesth. 2012;59(1):41-52.
- Noppens RR, Geimer S, Eisel N, et al. Endotracheal intubation using the C-MAC® video laryngoscope or the Macintosh laryngoscope: a prospective, comparative study in the ICU. Crit Care. 2012;16(3):R103.
- Sakles JC, Mosier JM, Chiu S, et al. Tracheal intubation in the emergency department: a comparison of GlideScope® video laryngoscopy to direct laryngoscopy in 822 intubations. J Emerg Med. 2012;42(4):400-405.
- Simpson GD, Ross MJ, McKeown DW, et al. Tracheal intubation in the critically ill: a multi-centre national study of practice and complications. Br J Anaesth. 2012;108(5):792-799.
- Cooper RM, Pacey JA, Bishop MJ, et al. Early clinical experience with a new videolaryngoscope (GlideScope) in 728 patients. Can J Anaesth. 2005;52(2):191-198.
- Healy DW, Maties O, Hovord D, et al. A systematic review of the role of videolaryngoscopy in successful orotracheal intubation. BMC Anesthesiol. 2012;12(12):32.
- Caplan RA, Posner KL, Ward RJ, et al. Adverse respiratory events in anesthesia: a closed claims analysis. Anesthesiology. 1990;72(5):828-833.
- Tinker JH, Dull DL, Caplan RA, et al. Role of monitoring devices in prevention of anesthetic mishaps: a closed claims analysis. Anesthesiology. 1989;71(4):541-546.
- Eichhorn JH. Documenting improved anesthesia outcome. J Clin Anesth. 1991;3(5):351-353.
- Eichhorn JH. Prevention of intraoperative anesthesia accidents and related severe injury through safety monitoring. Anesthesiology. 1989;70(4):572-577.
- Redan JA, Livingston DH, Tortella BJ, et al. The value of intubating and paralyzing patients with suspected head injury in the emergency department. J Trauma. 1991;31(3):371-375.
- Talucci R, Shaikh KA, Schwab CW. Rapid sequence induction with oral endotracheal intubation in the multiply injured patient. Am Surg. 1988;54(4):185-187.
- Tayal VS, Riggs RW, Marx JA, et al. Rapid-sequence intubation at an emergency medicine residency: success rate and adverse events during a two-year period. Acad Emerg Med. 1999;6(1):31-37.
- Rotondo MF, McGonigal MD, Schwab CW, et al. Urgent paralysis and intubation of trauma patients: is it safe? J Trauma. 1993;34(2):242-246.
- Norwood S, Myers MB, Butler TJ. The safety of emergency neuromuscular blockade and orotracheal intubation in the acutely injured trauma patient. J Am Coll Surg. 1994;179(6):646-652.
- Ligier B, Buchman TG, Breslow MJ, et al. The role of anesthetic induction agents and neuromuscular blockade in the endotracheal intubation of trauma victims. Surg Gynecol Obstet. 1991;173(6):477-481.
- Sakles JC, Laurin EG, Rantapaa AA, et al. Airway management in the emergency department: a one-year study of 610 tracheal intubations. Ann Emerg Med. 1998;31(3):325-332.
- Klinger JR. Hemodynamics and positive end-expiratory pressure in critically ill patients. Crit Care Clin. 1996;12(4):841-864.
- Milligan KR, Beers HT. Vecuronium-associated cardiac arrest. Anaesthesia. 1985;40(4):385.
- Kim WY, Kwak MK, Ko BS, et al. Factors associated with the occurrence of cardiac arrest after emergency tracheal intubation in the emergency department. PLoS One. 2014;9(11):e112779.
- Ko BS, Ahn R, Ryoo SM, et al. Prevalence and outcomes of endotracheal intubation–related cardiac arrest in the ED. Am J Emerg Med. 2015;33(11):1642-1645.
- De Jong A, Rolle A, Molinari N, et al. Cardiac arrest and mortality related to intubation procedure in critically ill adult patients: a multicenter cohort study. Crit Care Med. 2018;46(4):532-539.
- Cook TM, Woodall N, Frerk C, et al. Major complications of airway management in the UK: results of the Fourth National Audit Project of the Royal College of Anaesthetists and the Difficult Airway Society. Part 1: anaesthesia. Br J Anaesth. 2011;106(5):617-631.
- Cook TM, Woodall N, Harper J, et al. Major complications of airway management in the UK: results of the Fourth National Audit Project of the Royal College of Anaesthetists and the Difficult Airway Society. Part 2: intensive care and emergency departments. Br J Anaesth. 2011;106(5):632-642.
- Sanborn KV, Castro J, Kuroda M, et al. Detection of intraoperative incidents by electronic scanning of computerized anesthesia records. Comparison with voluntary reporting. Anesthesiology. 1996;85(5):977-987.
- Katz RI, Lagasse RS. Factors influencing the reporting of adverse perioperative outcomes to a quality management program. Anesth Analg. 2000;90(2):344-350.
- Over DC, Pace NA, Shearer VE, et al. Clinical audit of anaesthesia practice and adverse peri-operative events at Parkland Memorial Hospital, Dallas, Texas. Eur J Anaesthesiol. 1994;11(3):231-235.
- Goldman SA, Kennedy DL, Graham DJ, et al. The clinical impact of adverse event reporting. MedWatch, Center for Drug Evaluation and Research, FDA. www.nvic.org/?CMSTemplates/?NVIC/?pdf/?FDA/?vaers-medwatch-1996.pdf. October 1996. Accessed June 27, 2018.
- Jaber S, Jung B, Corne P, et al. An intervention to decrease complications related to endotracheal intubation in the intensive care unit: a prospective, multiple-center study. Intensive Care Med. 2010;36(2):248-255.


Leave a Reply
You must be logged in to post a comment.